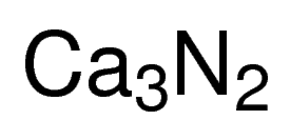Calcium Nitride
Calcium nitride, referred to as calcium azanidylidenecalcium in IUPAC nomenclature, is an inorganic compound with constituent elements calcium and nitrogen and represented by the chemical formula Ca3N2 [1]. It has different isomorphous forms, out of which α-calcium nitride is commonly found [2].
Calcium Nitride Identification |
|
| CAS Number | 12013-82-0 [3, 4] |
| PubChem CID | 3387080 [1] |
| ChemSpider ID | 21171155 [4] |
| EC Number | 234-592-9 [3] |
| RTECS Number | N/A [3] |
| InChI Key | XCNGEWCFFFJZJT-UHFFFAOYSA-N [1] |
How is Calcium Nitride Prepared
It can be synthesized when calcium is heated at an elevated temperature in the presence of air, which is represented by the following direct reaction of the elements [2]:
3Ca + N2 → Ca3N2
The calcium nitride formed can be easily identified, as it reacts with moisture or water to produce calcium hydroxide and ammonia [2]:
Ca3N2 + 6H2O → 3Ca(OH)2 + 2NH3
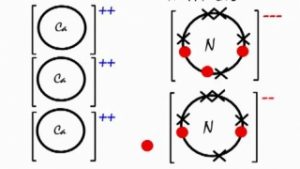
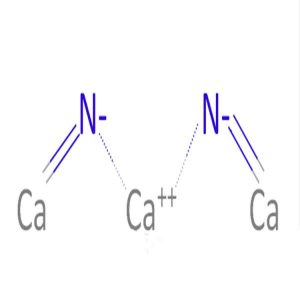
Calcium Nitride Ionic Bond
Properties and Characteristics of Calcium Nitride
General Properties |
|
| Molar Mass/Molecular Weight | 148.248 g/mol [1] |
Physical Properties |
|
| Color and Appearance | Reddish-brown crystalline powder [2, 3] |
| Melting Point | 1,195 °C, 2,183 °F [5] |
| Boiling Point | N/A [3] |
| Density | 2.63 g cm-3 [3] |
| State of matter at room temperature (solid/liquid/gas) | Solid [3] |
| Solubility | N/A |
| Solubility in Water | Decomposes in H2O |
Atomic Properties |
|
| Crystal Structure | Cubic [5] |
Uses
- Ca3N2 can be used for obtaining reactive nitride ions [5].
- Calcium hydride (a desiccant) can be produced by heating calcium nitride with hydrogen at temperatures above 350 °C [2].
Is it Toxic
Repeated contact or overexposure to Ca3N2 has been linked to eye damage and skin burns [6]. It is a flammable solid and should be kept away from sparks, heat, open flames, and water, because of possible dangerous reactions [6].
- References
- Calcium Nitride (Ca3N2) – Pubchem.ncbi.nlm.nih.gov
- Calcium Nitride – Revolvy.com
- Calcium Nitride – Americanelements.com
- Calcium bis(azanidylidenecalcium) – Chemspider.com
- Calcium Nitride Properties – Chembk.com
- Calcium Nitride – Chemicalbook.com