Combustion Reaction
What is a Combustion Reaction?
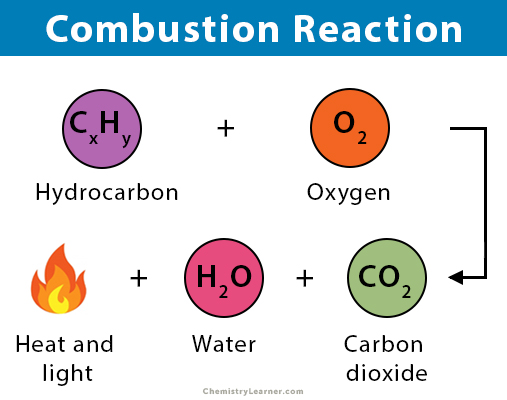
A combustion reaction is an exothermic chemical reaction between substances, usually including oxygen gas and accompanied by the generation of heat, energy, and light (flame). The products of a combustion reaction depend on the combusted substance. When metals and nonmetals burn in the presence of oxygen, they will give off their corresponding oxides. On the other hand, hydrocarbons give off carbon dioxide and water upon combustion.
A hydrocarbon is a compound made up solely of carbon and hydrogen. Hydrocarbons are the primary constituent of fossil fuels, namely natural gas, petroleum, and coal. They are used as fuel because their combustion releases enormous amounts of energy. Since different hydrocarbons have different ratios of hydrogen to carbon, they produce different ratios of water to carbon dioxide [1-6].
General Equation
The general equation for combustion reaction of metals and nonmetals is:
x M + (y/2) O2 → MxOy
The general equation for the combustion of hydrocarbon is [1]:
CxHy + (x+y/4) O2 → x CO2 + (y/2) H2O
Complete and Incomplete Combustion Reactions
Complete combustion reactions, also known as clean combustion reactions, is the complete oxidation of the fuel. Such reactions often liberate only water and carbon dioxide as the products, apart from heat and light. An example could be the combustion of propane in the presence of oxygen.
Incomplete combustion reactions are the combustion reactions that involve the formation of by-products such as ash and soot. This reaction happens when the supply of oxygen is inadequate, and carbon and carbon monoxide are produced. An example could be the combustion of propane in an environment where the oxygen is deficient [7].
How to Identify a Combustion Reaction?
A combustion reaction is a reaction in which one of the reactants is oxygen, and the other reactant is usually a hydrocarbon. The products are carbon dioxide and water, accompanied by heat and light (flame). The products are either complete or incomplete, in which case, carbon and carbon monoxide results. Inorganic substances can also burn in the presence of oxygen to give vapors of oxides [6].
Balancing Combustion Reaction [7]
Method 1: Balancing it directly.
Consider the combustion of the hydrocarbon propane (C3H8) in the presence of oxygen (O2), resulting in carbon dioxide (CO2) and water (H2O).
C3H8 (g) + O2 (g) → CO2 (g) + H2O (g)
We start with the carbon atom. We note that there are three atoms on the left-hand side of the equation and one on the right. So, we multiply CO2 by 3.
C3H8 (g) + O2 (g) → 3 CO2 (g) + H2O (g)
Then, we move to the hydrogen atom. We note that there are eight atoms on the left and two on the right. So, we multiply H2O by 4.
C3H8 (g) + O2 (g) → 3 CO2 (g) + 4 H2O (g)
Finally, we focus on oxygen. We find that there are ten atoms on the right and two on the left. So, we multiply O2 by 5.
C3H8 (g) + 5 O2 (g) → 3 CO2 (g) + 4 H2O (g)
This equation is now a balanced equation representing the combustion of propane.
Method 2: Using the general equation
We can also use the general equation to balance the combustion reaction equation.
CxHy + (x+y/4) O2 → x CO2 + (y/2) H2O
Here, x = 3 and y = 8.
C3H8 (g) + 5 O2 (g) → 3 CO2 (g) + 4 H2O (g)
Examples of Combustion Reaction
There are a few examples of combustion reactions of metals, nonmetals, and hydrocarbons. The reactions, along with the formulas of the compounds, are given below [1-10].
Combustion of Metals and Nonmetals
Metals and nonmetals combine with oxygen to give their corresponding oxides. These oxides are released in the atmosphere as gases. Some examples are as follows:
- Hydrogen (H2) reacts with oxygen (O2) to give water vapor (H2O).
2 H2 (g) + O2 (g) → 2 H2O (g)
- Carbon or coal (C) burns with oxygen to give and carbon monoxide (CO) and carbon dioxide (CO2)
2 C (s) + O2 (g) → 2 CO (g)
C (s) + O2 (g) → CO2 (g)
- Sulfur (S) burns with oxygen to give carbon dioxide (SO2)
S (s) + O2 (g) → SO2 (g)
- Magnesium (Mg) burns in oxygen (O2) to give magnesium oxide (MgO)
2 Mg (s) + O2 (g) → MgO (g)
- Combustion of nitrogen (N2) with oxygen results in nitric oxide (NO) and nitrogen dioxide (NO2)
N2 (g) + O2 (g) → 2 NO (g)
N2 (g) + 2 O2 (g) → 2 NO2 (g)
Combustion of Hydrocarbons
Hydrocarbons are an organic compound primarily made up of carbon and hydrogen. They are an excellent source of fuel as they ignite easily in the presence of oxygen to give carbon dioxide (CO2) and water (H2O).
- Methane (CH4)
CH4 (g) + 2 O2 (g) → CO2 (g) + 2 H2O (g)
- Ethane (C2H6)
2 C2H6 (g) + 7 O2 (g) → 4 CO2 (g) + 6 H2O (g)
- Butane (C4H10)
2 C4H10 (g) + 13 O2 (g) → 8 CO2 (g) + 10 H2O (g)
- Hexane (C6H14)
2 C6H14 (g) + 13 O2 (g) → 12 CO2 (g) + 3 H2O (g)
- Octane (C8H18)/gasoline
2 C8H18 (g) + 25 O2 (g) → 16 CO2 (g) + 18 H2O (g)
- Methanol
2 CH3OH (l) + 3 O2 (g) → 2 CO2 (g) + 4 H2O (g)
- Ethylene
C2H4 (g) + 3 O2 (g) → 2 CO2 (g) + 2 H2O (g)
- Acetone
C3H6O (l) + 4 O2 (g) → 3 CO2 (g) + 3 H2O (g)
- Naphthalene
C10H8 (s) + 12 O2 (g) → 10 CO2 (g) + 4 H2O (g)
- Benzoic acid
2 C6H5CO2H (s) + 15 O2 (g) → 14 CO2 (g) + 6 H2O (g)
Examples and Uses of Combustion Reaction in Everyday Life
There are a few examples and uses of combustion reactions in real life [10].
- Heating homes using wood or coal
- Running vehicles using petrol or diesel
- Lighting candles, which is made from paraffin wax, a hydrocarbon fuel
- Cooking food on a stovetop using natural gas or liquefied petroleum gas (LPG)
- Lighting a lighter, which consists of butane, a highly flammable hydrocarbon fuel
- Lighting a matchstick due to the presence of sulfur
- Production of energy from coal, oil, and natural gas in thermal power plants generates electricity
- Bursting of firecrackers, which contain many flammable chemicals
- Combustion of rocket propellent, which contains many hydrocarbons and its derivatives
FAQ
Ans. No. Not all combustion reactions are redox reactions.