E2 Reaction
What is E2 Reaction
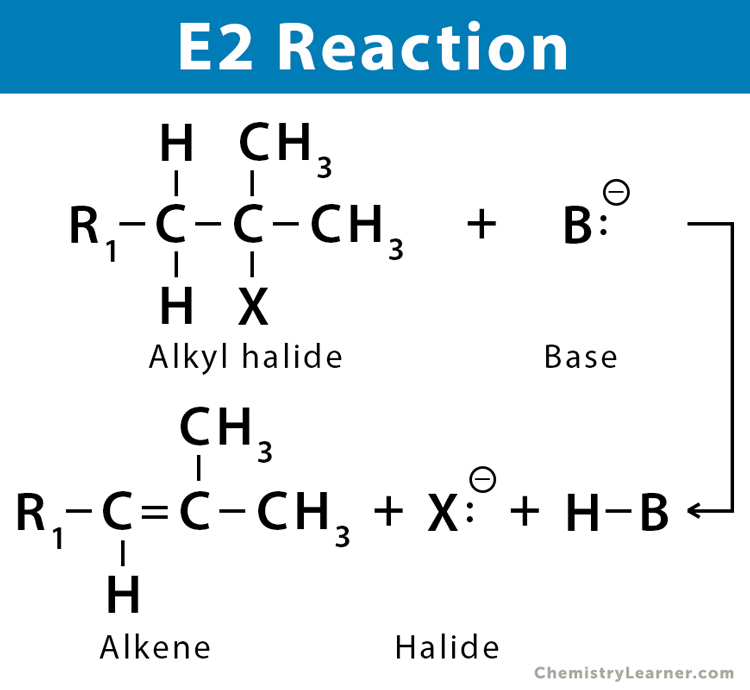
E2 reaction is a type of elimination reaction. A beta-hydrogen and a leaving group are removed from a compound (substrate) to form a double bond in a single concerted step. The removal usually takes place in the presence of a strong base that removes the proton. The rate of reaction is proportional to the concentration of both the substrate and the base. Therefore, a good leaving group is required because it is involved in the rate-determining step. In addition, the leaving group must be coplanar in order to form a pi bond. The E2 reaction is common in alkyl halide and is similar to the SN2 reaction [1-7].
Characteristics of E2 Reaction
- Also known as bimolecular elimination
- One step removal mechanism process (concerted)
- Carbon-leaving group and carbon-hydrogen bonds break off simultaneously
- Carbon goes from sp3 to sp2 hybridization state
- Reaction rate is proportional to both the substrates and the base – second-order kinetics (R = k [substrate][base])
- Better leaving group leads to faster rates
- Alkoxide (RO–) and hydroxide (OH–) bases are generally used
- Polar aprotic solvents increase the rate
- Both hydrogen and leaving group must be on the same plane in order for the double bond to form (coplanar)
- Stereoselective and regioselective
Regioselectivity and Stereochemistry of E2 Reaction
Regioselectivity
In an E2 reaction, there can be more than one product. However, the most stable product is favored. Therefore, according to Zaitsev’s rule, the product with the most substituted carbon in the C=C pi bond will be thermodynamically the most stable (Zaitsev product).
The reactivity of the carbocation towards the E2 reaction is as follows:
Tertiary > Secondary > Primary > Methyl
Stereochemistry
The bond connecting the leaving group to the alpha-carbon and the bond connecting the hydrogen to the beta-carbon must be in the same plane. This orientation allows for the proper overlapping of the two carbon orbitals and facilitates the formation of the pi-bond. Furthermore, the hydrogen and the leaving group must be in anti-position to each other, i.e., they must be in an anti-periplanar geometry. This orientation allows the transition state of the reaction to be stable and lower in energy. Therefore, overall, the hydrogen and the leaving must be in anti-coplanar conformation. Because of the anti-coplanar conformation, one stereoisomer (E isomer) will be produced preferably over the other [8].
Examples of E2 Reaction [1-7]
1. Dehydrohalogenation of Alkyl Halides
1. In the presence of potassium ethoxide (C2H5O– K+) in ethanol (C2H5OH), isobutyl bromide (C4H9Br) converts into isobutene (C4H8).
C4H9Br + C2H5O– K+ → C4H8 + C2H5OH + KBr
2. In the presence of potassium ethoxide (C2H5O– K+) in ethanol (C2H5OH), 2-bromopropane (C3H7Br) gives propene (C3H6).
C3H7Br +→ C3H6 + C2H5OH + KBr
2. Dehydrohalogenation of Cyclic Compound
In the presence of alcoholic potassium hydroxide (KOH/C2H5OH), 2-methyl-1-chlorocyclohexane (C7H13Cl) gives 1-methylcyclohexene (C7H12).
C7H13Cl + + C2H5O– K+ → C7H12 + C2H5OH + KBr
Mechanism of E2 Reaction
The mechanism of the E2 reaction is a single-step concerted process with one transition state. First, the base removes the proton from the beta-carbon, and the electron pair in the beta-carbon forms a pi-bond with the alpha carbon. Then, finally, the leaving group comes off from the alpha-carbon with an electron pair [1-7, 9-12].
Reactivity of Carbocation
The reactivity of the carbocation towards the E2 reaction is as follows:
Tertiary > Secondary > Primary > Methyl