Electrochemical Series
The electrochemical series is a list that provides insights into the behavior of chemical elements and their compounds in various chemical reactions. It ranks elements based on their standard electrode potentials, correlating with their tendency to gain or lose electrons. This series effectively arranges elements in order of increasing or decreasing electrochemical activity, depending on their electropositive or electronegative nature. [1-4]
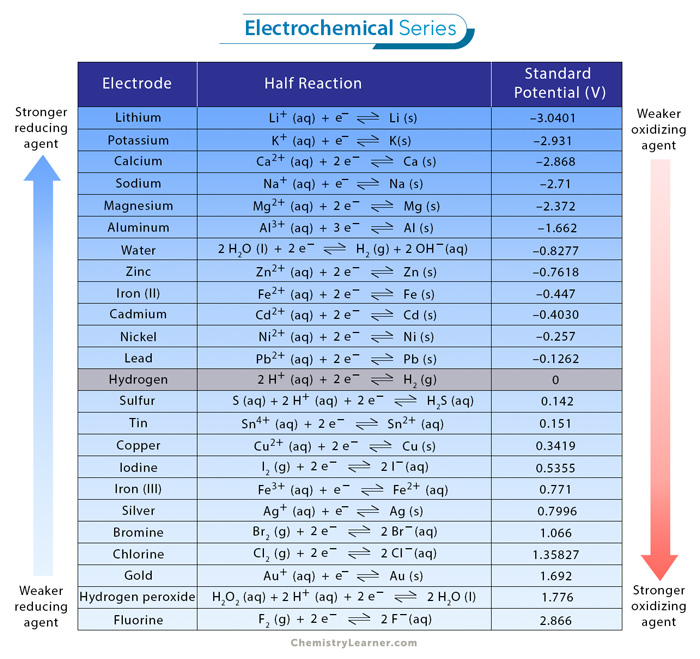
Electrochemical Series Chart
The electrochemical series is primarily evaluated by the elements’ ability to undergo reduction (gain electrons) or oxidation (lose electrons) during chemical processes. Their standard electrode potentials (E°) determine the placement of elements in the electrochemical series chart, often measured relative to the standard hydrogen electrode (SHE).
Elements with more negative electrode potentials are higher in the series, indicating a higher tendency to lose electrons, making them good reducing agents. Conversely, elements with more positive electrode potentials are at the bottom, representing a greater likelihood of gaining electrons and functioning as oxidizing agents. The placement of elements in the chart reflects their intrinsic reactivity and willingness to participate in redox reactions. [5,6]
Applications of the Electrochemical Series
The electrochemical series plays a vital role in various areas of chemistry and industry. Here, we explore some of its most prominent applications: [1-4]
Corrosion and Rust Prevention
One of the most widespread applications of the electrochemical series is in corrosion and rust prevention. When metals come into contact with moisture and oxygen, they are susceptible to corrosion. The electrochemical series helps us understand the likelihood of corrosion for different metals. By knowing the relative positions of metals in the series, we can predict which ones are more prone to oxidation and corrosion. This knowledge is fundamental in selecting suitable materials for construction, infrastructure, and machinery, ensuring longevity and safety.
Predicting the Feasibility of Redox Reaction
When two elements are involved in a chemical reaction, the one higher in the series will tend to undergo oxidation, while the one lower in the series will undergo reduction. The difference in their positions on the chart helps determine the direction of electron flow in a redox reaction. Understanding this direction is critical for predicting the spontaneity of chemical reactions and the feasibility of various electrochemical processes, such as the operation of batteries and galvanic cells.
In an electrolytic cell, the anode is the oxidation site, while the cathode is where reduction occurs. The known cell voltages play a crucial role in understanding electron movement, as the standard emf of the cell (Ecell) quantifies the potential of the reducing agent undergoing oxidation and the oxidizing agent undergoing reduction.
The following equation expresses this relationship:
E°cell = E°ox + E°red
Where E°ox is the standard oxidation potential, and E°red is the standard reduction potential.
When we utilize the Gibbs free energy equation, a positive value of E°cell implies a negative ΔG, indicating the feasibility of the reaction. The cell reaction occurs spontaneously and generates electrical energy. However, if E°cell is negative, the spontaneous reaction is not feasible. Let us test this by taking an example of corrosion.
In the experiment involving steel immersed in freshwater, the half-cell reactions for iron and neutral water are:
1. O2 + 2 H2O + 4 e— → 4 OH— with E° = +0.40 V
2. Fe2+ + 2 e— → Fe with E° = -0.45 V
When assessing steel corrosion, it is imperative to designate the iron reaction as the anode. To achieve this, invert the direction of the reaction, ensuring that iron is oxidized. It leads to the following values:
E°ox = +0.45 V
E°red = +0.40 V
Subsequently, by employing the equation:
E = E°ox + E°red
= (0.45) V + (0.40) V = +0.85 V
Given the positive E value, ΔG hs is negative, indicating the feasibility of steel corrosion in this context.
Galvanic Cells and Batteries
The electrochemical series is at the heart of galvanic cells and batteries. The potential difference between two half-reactions in a galvanic cell generates electrical energy. The electrochemical series helps select the right combination of electrode materials to create the desired voltage and current. In batteries, the series is used to design and optimize the electrochemical reactions that produce electrical energy, leading to the development of batteries with varying capacities, sizes, and applications in electronics, transportation, and renewable energy storage.
Electroplating and Metal Finishing
Electroplating is widely used to coat objects with a different metal layer for various purposes, including aesthetics and protection. The electrochemical series is instrumental in selecting the appropriate materials for the anode and cathode during electroplating. Manufacturers can achieve high-quality and durable electroplated finishes on various products by choosing metals with the desired electrochemical potential.
Metallurgy and Extraction of Metals
Metallurgy and the extraction of metals from ores depend on the reactivity and electrochemical properties of elements. The electrochemical series helps metallurgists select the most efficient and cost-effective methods for extracting metals from their ores. It guides decisions regarding which reducing agents to use and which metals can be easily separated from their compounds. This knowledge is crucial in industries ranging from steel production to precious metal mining.