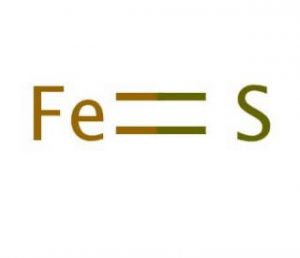Iron(II) Sulfide
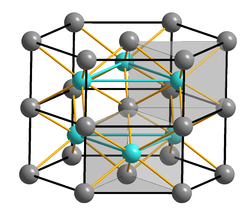
Iron(II) sulfide, also popular as iron sulfide or ferrous sulfide, is an inorganic, water-insoluble compound represented by the chemical formula FeS [1, 2]. In IUPAC nomenclature, it is called sulfanylideneiron [1]. It naturally occurs as the minerals troilite and pyrrhotite [3]. FeS has the nickel arsenide structure, with trigonal prismatic sulfides and octahedral iron centers [5].
Iron(II) Sulfide Identification |
|
| CAS Number | 1317-37-9 [1] |
| PubChem CID | 14828 [1] |
| ChemSpider ID | 8466211 [2] |
| EC Number | 215-268-6 [1] |
| MDL Number | MFCD00011013 [1] |
| InChI Key | MBMLMWLHJBBADN-UHFFFAOYSA-N [1] |
Iron(II) Sulfide Composition and Synthesis
When iron and sulfur are mixed and heated in a test tube, it undergoes an exothermic reaction to produce iron(ii) sulfide [6].
Fe + S → FeS
Reaction with Hydrochloric Acid
Iron sulfide reacts with hydrochloric acid to yield ferrous chloride and hydrogen sulfide, as indicated by the following reaction:
FeS + 2 HCl → FeCl2 + H2S
Properties and Characteristics of Iron(II) Sulfide
General Properties |
|
| Molar Mass/Molecular Weight | 87.905 g/mol [1] |
Physical Properties |
|
| Color and Appearance | Gray to brownish-black rods, lumps, or granular powder, colorless when pure [1] |
| Odor | Odorless [8] |
| Melting Point | 1194 °C, 2181 °F [1] |
| Boiling Point | Decomposes [1] |
| Density | 4.75 g cm-3 [1] |
| State of matter at room temperature | Solid [1] |
| Solubility | Reacts in acids with the production of hydrogen sulfide, insoluble in nitric acid [1] |
| Solubility in Water | 0.00062 g/100 cc at 18 °C [1] |
| Magnetic Susceptibility (χ) | 1074 X 10-6 cm3/mol |
Atomic Properties |
|
| Crystal Structure | Octahedral |
Uses
- As a pigment in ceramics, hair dyes, and glass containers [1, 7].
- Treating exhaust gases and lessening heavy metal pollution [1].
- To synthesize hydrogen sulfide in the laboratory [1].
Is It Safe
Iron sulfide can cause irritation to your skin, eyes, lungs, and gastrointestinal tract when inhaled [1]. It is known to cause slight toxicity upon ingestion [8].
- References
- Ferrous Sulfide – Pubchem.ncbi.nlm.nih.gov
- Iron(II) Sulfide – Chemspider.com
- Ferrous Sulfide – Chemicalbook.com
- Iron(II) Sulfide – Americanelements.com
- Structures Related to NaCl and NiAs – Chem.libretexts.org
- Iron and Sulfur Reaction – Rsc.org
- Iron(II) Sulfide – Hazmap.nlm.nih.gov
- Iron(II) Sulfide – Mccsd.net