Home / Inorganic Chemistry / Magnesium Nitrite
Magnesium Nitrite
Table Of Contents
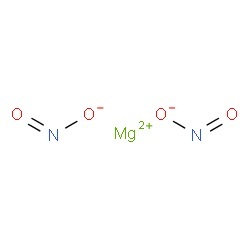
Magnesium nitrite represented by the chemical formula Mg(NO2)2 or MgN2O4 that bears the IUPAC name magnesium dinitride [1] is a white powder at room temperature that is soluble in water and alcohol [3, 4]. It is a magnesium salt and an ionic compound [1]. It should be protected from moisture to prevent it from decomposing [4].
Magnesium Nitrite Identification |
|
| CAS Number | 15070-34-5 [1] |
| PubChem CID | 10197667 [1] |
| ChemSpider ID | 8373167 [2] |
| UNII | 05MVC8T5JN [1] |
How to Make It
Magnesium nitrite can be prepared by reacting magnesium hydroxide with nitrous acid [3].
Mg(OH)2 + 2HNO2 = Mg(NO2)2 + 2H2O
It can also be made by a reaction between cobalt nitrite and magnesium [3].
Co(NO2)2 + Mg = Mg(NO2)2 + Co
Properties and Characteristics of Magnesium Nitrite
General Properties |
|
| Molar mass/molecular weight | 116.315 g/mol [1] |
Physical Properties |
|
| Color/appearance | White hygroscopic crystals [3, 4] |
| Melting point/freezing point | Unknown [2] |
| Boiling point | Unknown [2] |
| Density | Unknown [2] |
| State of matter at room temperature (normal phase) | Solid [3] |
Magnesium Nitrite Uses
- In research work in laboratories [6]. It is only available in small amounts commercially and is unstable limiting its use [4].
Is It Dangerous
Though it is generally non-hazardous, it may pose a health risk if ingested [4, 5].
- References
- Magnesium Nitrite – Pubchem.ncbi.nlm.nih.gov
- Magnesium Nitrite – Chemspider.com
- Magnesium Nitrite – Endmemo.com
- Magnesium Nitrite – Encyclopedia of the Alkaline Earth Compounds
- Nitrate in foods: harmful or healthy? – ltschem.com
- Magnesium Nitrite, Mg(NO2)2 – Magnesium.atomistry.com