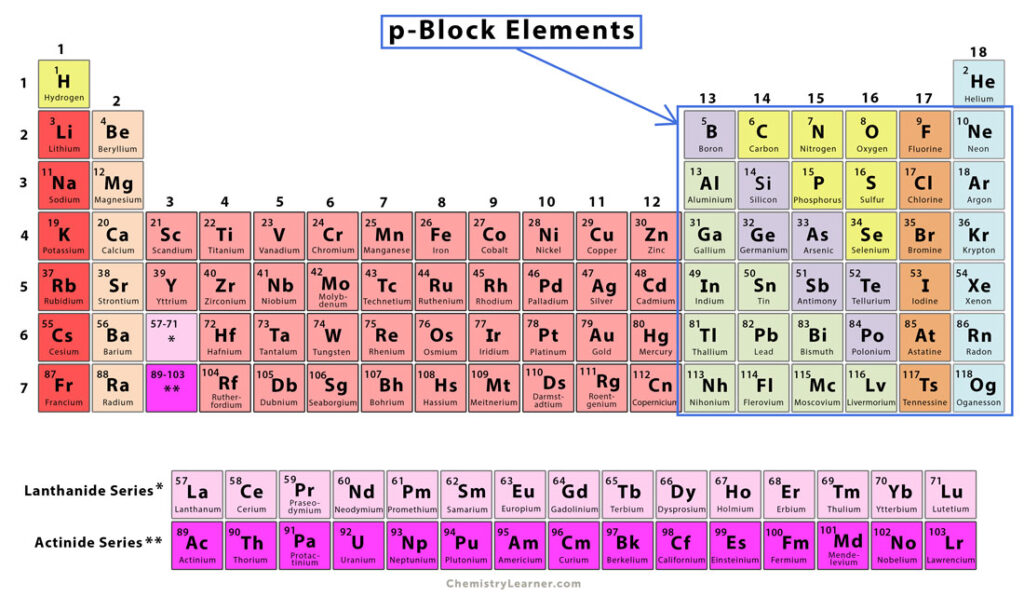
p-Block Elements
p-block elements are those periodic table elements whose last valence electron goes into any of the three p orbitals. Since there are three p orbitals, each occupying up to two electrons, the maximum number of electrons in the p subshell is six. Therefore, there are six groups of p-block elements [1-4].
Location
p-block elements are located on the right-hand side of the periodic table. Their group numbers range from 13 to 18. Group 13 elements have one p-orbital electron, Group 14 elements have two, and so on. Despite lying in Group 18, helium is not considered a p-block element. It is a s-block element [1-4].
Characteristics
p-block elements consist of metals, nonmetals, and metalloids. Since the elements are spread over various groups, their properties vary considerably. The first row of p-block elements obeys the octet rule. The rest of them are hypervalent, which means they can accommodate more than eight electrons in their valence shell. The p-block elements have multiple oxidation states, usually varying by multiples of two [1-4].
Electron Configuration
Valence Shell Electron Configuration
| Group Number | Group Name | |
|---|---|---|
| Group 13 | Boron family | ns2np1 |
| Group 14 | Carbon family | ns2np2 |
| Group 15 | Nitrogen family | ns2np3 |
| Group 16 | Oxygen family (The chalcogens) | ns2np4 |
| Group 17 | Halogen family | ns2np5 |
| Group 18 | Nobel gases | ns2np6, except helium, whose electron configuration is 1s2 |
Properties [1-4]
- The p-block element’s penultimate shell consists of 2, 8, or 18 electrons.
- Except for inert gases, p-block elements have a wide range of oxidation states. They run from +n to (n-8). Here, n is the number of electrons in the outermost shell.
- The p-block elements have covalency, but higher members can have electrovalency. Highly electronegative elements, like fluorine, chlorine, and other halogens, display electrovalency by accepting electrons and forming anions. Some p-block elements also have coordinate valency.
- The non-metallic character of the elements increases from left to right over a period. It decreases from top to bottom down a group.
- Ionization energies increase from left to right across a period and decrease from top to bottom down a group. Because of the half-filled and fully-filled orbitals in the valence shell, members of groups 15 and 18 have very high ionization energies.
- The reducing tendency decreases from left to right across a period, whereas oxidizing nature increases. Halogens are strong oxidizers.
- Most p-block elements react with oxygen to form acidic oxides. The acidic character increases from left to right across a period.
- Unlike s-block elements, no p-block elements nor their salts impart a distinct color to a flame.
- Several p-block elements exhibit a phenomenon called allotropy. Carbon, silicon, phosphorus, sulfur, boron, germanium, tin, and arsenic are known to display this property.
- Many p-block elements display catenation properties. These include carbon, silicon, germanium, nitrogen, oxygen, and sulfur.