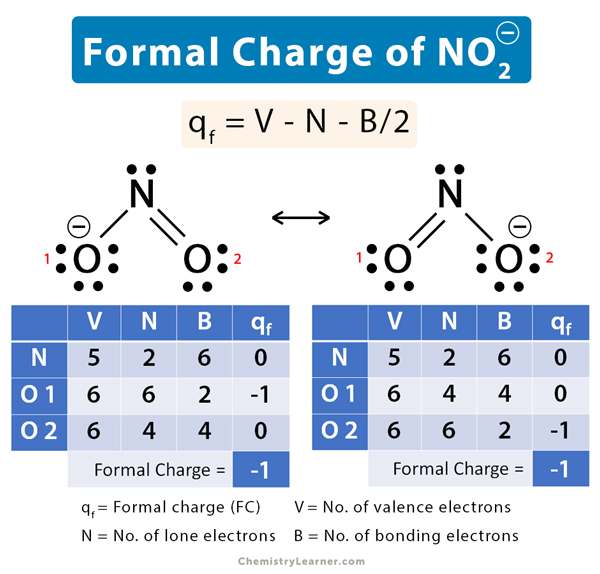
Nitrite Ion (NO2–) Formal Charge
In the nitrite ion (NO2–), a single covalent bond connects one oxygen (O) atom with the nitrogen (N) atom. Another oxygen atom is bonded to the nitrogen by a double covalent bond. There are two resonance structures.
O–-N=O ↔ O=N-O–
Let us calculate the formal charges on the atoms and determine the formal charge of NO2–.
i. O–-N=O
V = 5, N = 2, B = 6
Therefore,
qf = 5 – 2 – 6/2 = 0
Oxygen 1
V = 6, N = 6, B = 2
Therefore,
qf = 6 – 6 – 2/2 = -1
Oxygen 2
V = 6, N = 4, B = 4
Therefore,
qf = 6 – 4 – 4/2 = 0
The net formal charge is: 0 -1 + 0 = -1
ii. O=N-O–
Nitrogen
V = 5, N = 2, B = 6
Therefore,
qf = 5 – 2 – 6/2 = 0
Oxygen 1
V = 6, N = 4, B = 4
Therefore,
qf = 6 – 4 – 4/2 = 0
Oxygen 2
V = 6, N = 6, B = 2
Therefore,
qf = 6 – 6 – 2/2 = -1
The net formal charge is: 0 + 0 – 1 = -1
The two resonance structures of the nitrite ion form a hybrid with formal charge equal to -1.