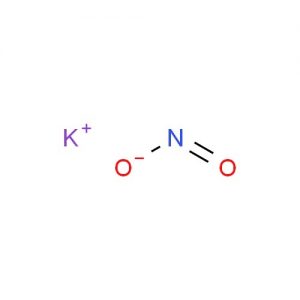
Potassium Nitrite
Potassium nitrite represented by the chemical formula KNO2 is a yellowish white crystalline and deliquescent powder that is soluble in water, alcohol and liquid ammonia [1]. It is a potassium salt and an ionic compound [1, 3]. KNO2 is a strong oxidizer and incompatible with strong acids, strong reducing agents, cyanides, ammonium salts and combustible materials [6].
Potassium Nitrite Identification |
|
| CAS Number | 7758-09-0 [1] |
| PubChem CID | 516910 [1] |
| ChemSpider ID | 22857 [2] |
| EC Number | 231-832-4 [1] |
How to Make It
Potassium nitrite can be prepared by heating potassium nitrate with lead.
KNO3 + Pb = KNO2 + PbO
If you want, you can use zinc or copper instead of lead [5].
Properties and Characteristics of Potassium Nitrite
General Properties |
|
| Molar mass/molecular weight | 85.103 g/mol [1] |
Physical Properties |
|
| Color/appearance | White or slightly yellow crystals [1] |
| Melting point/freezing point | 441°C, 825.8°F [1] |
| Boiling point | 537°C, 998.6°F [1] |
| Density | 1.9 g cm-3 [1] |
| State of matter at room temperature (normal phase) | Solid [1] |
| Heat capacity | 107.4 J/mol K [1] |
| Specific gravity | 1.915 [6] |
Chemical Properties |
|
| Flammability | Yes [4] |
| Solubility in water | 3120 g/l (at 25oC) [1] |
| pH | 8.40 (basic) [3] |
Prominent Reactions
A reaction between potassium nitrite and concentrated nitric acid produces potassium nitrate, nitrogen dioxide, nitric oxide and water [7].
2KNO2 + 2HNO3 = 2KNO3 + NO2 + NO + H2O
A saturated solution of KNO2 reacts with concentrated sulfuric acid to produce potassium sulfate, nitrogen dioxide, nitric oxide and water [8].
2KNO2 + H2SO4 = K2SO4 + NO2 + NO + H2O
Potassium nitrite reacts with concentrated hydrochloric acid to form potassium nitrate, potassium chloride, nitric oxide and water [9].
3KNO2 + 2HCl = KNO3 + 2KCl + 2NO + H2O
Potassium Nitrite Uses
- To make other chemicals, heat transfer salts [1, 6].
- In chemical analysis [1].
- As a food additive (preservative), in fertilizers [4].
- As an antidote to cyanide poisoning [4].
- As a vasodilator in the medical field [4].
- As an anti-scaling agent and corrosion inhibitor [6].
Is It Dangerous
It is a poison if ingested. It may cause explosion hazard if heated. The compound begins decomposing at 350°C and emits toxic fumes of potassium oxide. KNO2 in food can increase the risk of cancer [1, 4].
Potassium Nitrite Price
10 gram of the compound costs around $19.2 [6].
- References
- Potassium Nitrite – Pubchem.ncbi.nlm.nih.gov
- Potassium Nitrite – Chemspider.com
- What is the pH of a 0.25M solution of potassium nitrite? – Quora.com
- Potassium Nitrite – Chemicalbook.com
- Preparation of potassium nitrite – Prepchem.com
- Potassium Nitrite – Chemicalbook.com
- Potassium nitrite react with nitric acid – Chemiday.com
- Potassium nitrite react with sulfuric acid – Chemiday.com
- Potassium nitrite react with hydrogen chloride – Chemiday.com