Reaction Rate
A chemical reaction can be fast or slow depending upon many factors. For example, wood burning occurs quickly, and rust formation occurs slowly. The reaction rate or rate of reaction is the speed at which a chemical reaction proceeds. It is expressed as the concentration of the reactants consumed or products formed per unit time [1-4].
Rate of Reaction Formula
The general expression for the reaction rate is [1-9]
Rate = Change in Concentration/Time
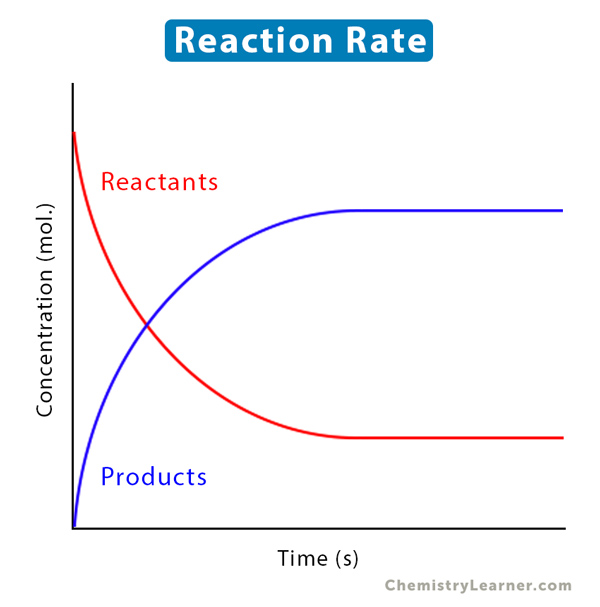
A chemical reaction can be simple or complicated. A simple reaction consists of a single reactant that forms a product. A complicated reaction consists of several reactants combining to form products.
Simple Reaction
Consider a simple reaction in which reactant A forms product B.
A → B
Then, the reaction rate (R) is given by
R = Decrease in concentration of A/Time
Or, R = Increase in concentration of B/Time
Since A is the only reactant and B is the only product, and mass is conserved in the reaction, the decrease in A’s concentration is equal to the increase in B’s concentration. Therefore,
R = Decrease in concentration of A/Time = Increase in concentration of B/Time
The above equation can be interpreted as the rate of disappearance of A being equal to the rate of appearance of B. The unit of reaction rate is moles per liter per second or mol L-1 s-1.
Average Reaction Rate
Since the concentrations of A and B change over time, it will be appropriate to calculate the average reaction rate. Suppose the A and B concentrations are recorded at time t1 and then at t2. The average reaction rate is given by
R = ([A]2 – [A]1)/(t2 – t1) = ([B]2 – [B]1)/(t2 – t1)
Where
[A]1: Concentration of A at time t1
[A]2: Concentration of A at time t2
[B]1: Concentration of B at time t1
[B]2: Concentration of B at time t2
The above equation can be simplified to
R = – Δ[A]/Δt = Δ[B]/Δt
The Greek symbol delta (Δ) indicates change, which can be experimentally measured. The negative sign implies that A’s concentration is decreasing over time, i.e., [A]2 < [A]1.
Instantaneous Reaction Rate
A disadvantage of using the average reaction rate is that it only gives the average over time. This value may differ for different time intervals. To consider this variation, we use the instantaneous reaction rate at a particular instant of time. Then, the equation is expressed in differential form as follows:
R = -d[A]/dt = d[B]/dt
Complicated Reaction
The reaction rate can be evaluated for reactions with multiple reactants and products. Such complicated reactions are typically performed in laboratories. Consider the following reaction:
aA + bB ⇄ cC + dD
Where a, b, c, and d are the stoichiometric coefficients of the reactants A and B and products C and D, respectively.
IUPAC’s Gold Book defines reaction rate (r) as follows:
r = -1/a d[A]/dt = -1/b d[B]/dt = 1/c d[C]/dt = 1/d d[D]/dt
The above equation is true for any reaction occurring in a closed system at constant volume without reaction intermediates formation.
Factors Affecting Rate of Reaction [1-9]
Several factors influence the reaction rate. The reaction rate is altered when we change the reaction conditions.
Temperature
According to kinetic theory, higher temperature makes the molecules travel faster. The number of molecular collisions increases since these molecules have already gained the activation energy required to overcome the barrier. As a result, the reaction proceeds faster at a higher temperature than lower temperature.
However, there are some reactions whose rate decreases with temperature, while a few other reactions are not affected at all.
Concentration
According to the law of mass action, the reaction rate is directly proportional to the concentration. It increases as the concentration increases and decreases as the concentration decreases.
Pressure
Pressure affects the reaction rate, especially for gases. When the pressure is increased, the molecules are confined to a smaller space. The probability of collision increases, resulting in product formation and a higher reaction rate. The opposite is also valid. When the pressure is decreased, the molecules can move freely. The number of collisions reduces, decreasing the reaction rate.
Catalyst
The role of a catalyst is to lower the activation energy and accelerate a chemical reaction. Hence, the presence of a catalyst will increase the reaction rate. You may read our article on catalytic reactions to learn more.
Light or Electromagnetic Radiation
The presence of light or other electromagnetic radiation will increase the reaction rate. The reason is that electromagnetic radiation carries energy, which is then imparted to the molecules. The energized molecules collide more often, resulting in a faster reaction.
Physical State and Surface Area
If the reactants are in different physical states, the reaction rate will be limited by the surface area in contact. For example, if a solid metal reacts with a gaseous substance, only the atoms on the surface will react. It means that a larger surface area results in a higher value of the reaction rate.