Strontium Iodide
Strontium iodide, also called strontium diiodide, is a salt containing strontium and iodine [1, 2]. Being a water-soluble, deliquescent compound, it absorbs moisture from the atmosphere and becomes physically changed slightly [2]. It produces strontium oxide and iodine when heated at high temperatures in the presence of air [2].
Identification
CAS Number: 10476-86-5 [2, 3]
PubChem CID: 25304 [1, 2]
ChemSpider ID: 23637 [3]
EC Number: 233-972-1 [2, 3]
RTECS Number: WK9275000 [2, 4]
Formula
The molecular formula of its anhydrous form is I2Sr [1], while the linear formula is SrI2 [4]. Its hexahydrate form has the chemical formula SrI2.6H2O [2].
Structure
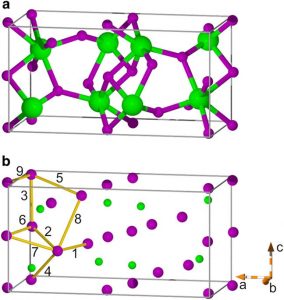
Strontium iodide has an orthorhombic crystal structure [2].
Properties
Here we share some common physical properties of this ionic compound.
Appearance
It appears as a colorless or white crystalline solid [2, 4]
Odor
Strontium iodide is odorless [5].
Molar Mass
The mass of one mole of strontium iodide is 341.43 g/mol [1, 2, 4]
Melting Point
The compound melts at a temperature of 507-645 °C (780-918 K, 945-1193 °F) [2, 4]
Boiling Point
It boils at a temperature of 1773 °C (2046 K, 3223 °F) [2, 4].
Density
The density of its anhydrous form is 4.55 g/cm3, and its hexahydrate form is 4.40 g/cm3 [2, 4].
Solubility
It is highly soluble in water at room temperature and sparingly soluble in ethanol, with a solubility value of 177 g/100 ml at 20 °C and 3.1 g/100 ml at 4 °C, respectively [2].
How to make Strontium Iodide
Its preparation involves the reaction of strontium carbonate (SrCO3) with hydroiodic acid (HI) [2].
SrCO3 + 2HI → SrI2 + H2O + CO2
The strontium iodide produced can be easily identified because it becomes yellow upon exposure to air [2].
Chemical Reactions with Other Compounds
Lithium Sulfide and Strontium Iodide
When SrI2 reacts with Li2S, the compounds undergo a double displacement reaction to form SrS and LiI [6]. The chemical equation is given below:
Li2S + SrI2 → 2LiI + SrS
Potassium Sulfate and Strontium Iodide
The precipitation reaction involving potassium sulfate and strontium iodide is given below:
K2SO4 + SrI2 → 2KI + SrSO4
Uses
- Because of its high density, atomic number, scintillation light yield, and optical clarity, Europium-doped SrI2 is used as a scintillator for detecting gamma radiation [2].
- Anhydrous strontium iodide is used as a replacement for potassium iodide in medicines [2].
- Large-diameter strontium iodide crystals are now commercially grown with the vertical Bridgman technique [2, 7].
Health and Safety Information
Although the toxicological properties of SrI2 have not been extensively studied, compounds of strontium are known to be slightly toxic [5]. Prolonged exposure through inhalation may cause headaches, tight chest, nose and respiratory system irritation, and lacrimation [5]. If ingested, it may cause gastric irritation, diarrhea, abdominal pain, and burns to the mouth [5]. Moreover, it may act as a strong irritant to the eye and skin, causing redness, black spots, boils, pimples, blisters, and hives [5].
First Aid Measures
If a victim has been exposed to large amounts of strontium iodide, the following measures should be taken:
Ingestion: Seek medical attention immediately and avoid inducing vomiting [5].
Inhalation: Remove the affected person to fresh air and keep him warm [5]. The victim should be immediately taken to a medical practitioner [5].
Eyes: Flush the victim’s eyes with lukewarm water for more than 15 minutes and seek emergency medical treatment [5].
Skin: After removing the contaminated clothes, brush the material off the skin and wash the affected area using mild soap and water [5]. Quickly take the person to a doctor to avoid acute irritation [5].
Safety Measures
For breathing protection, a NIOSH-approved respirator should be worn [5]. The compound should be kept in an inert gas and dry box to avoid contact with moisture [5]. Other protective equipments like an apron, lab coat, impervious gloves, and safety goggles are required for the safety of the skin and eyes [5].
Strontium Iodide Availability
Hangzhou Dayangchem Co. Ltd., IBIS Chemie International, Amadis Chemical Co. Ltd., Chemos GmbH & Co. KG, and Santa Cruz Biotechnology Inc. are some of the renowned suppliers of SrI2 globally [9].
References
- Strontium Iodide (SrI2) – Pubchem.ncbi.nlm.nih.gov
- Strontium Iodide – Wikipedia.org
- WK9275000 – Chemspider.com
- Strontium Iodide – Americanelements.com
- Strontium Iodide Material Safety Data Sheet – Espimetals.com
- Chemical Equation Balancer – Chemicalaid.com
- Crystal growth of large diameter strontium iodide scintillators using in Situ stoichiometry monitoring – Researchgate.net