Ionization Energy
What is Ionization Energy
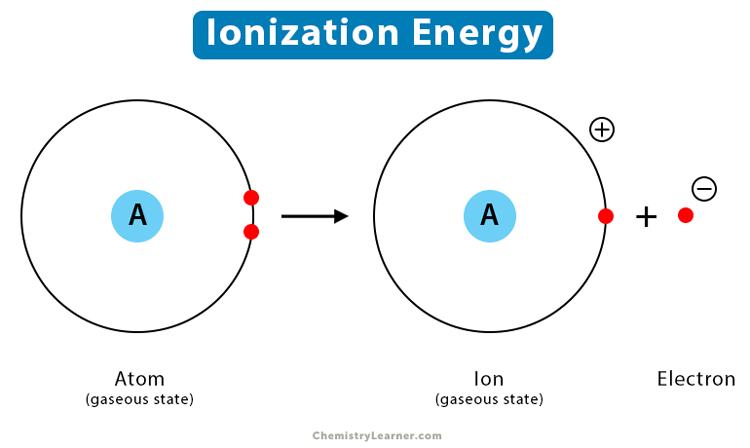
Ionization energy is the minimum energy required to remove a loosely bound electron of an atom or molecule in the gaseous state. It measures the capability of an atom to lose an electron during a chemical reaction. It is opposite to electron affinity [1-4].
What Happens During Ionization
During the ionization process, the applied energy removes an electron from the valence shell. It transforms the atom or molecule into a monovalent cation. Since energy is added to the system, the process is endothermic, and the ionization values are positive. The unit of ionization energy is kJ/mol, and the symbol is I. Since only one electron is removed, the ionization energy is also known as the first ionization energy.
How to Write a Chemical Equation for First Ionization Energy
The chemical reaction for the first ionization energy is given by [1-3],
X (g) → X+ (g) + e–
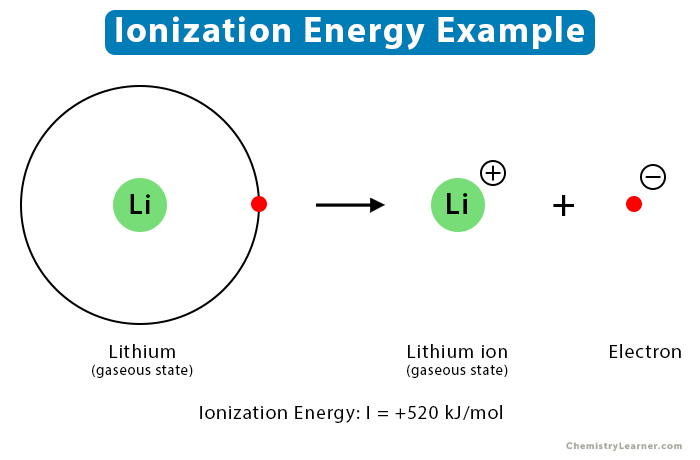
Example: Consider the ionization of lithium (Li) atom, which has a single electron in its 2s valence shell. In order to remove this electron, 520 kJ/mol of energy is required. The chemical equation is shown as follows.
Li (g) → Li+ (g) + e–
Therefore, ionization energy of lithium: I = +520 kJ/mol
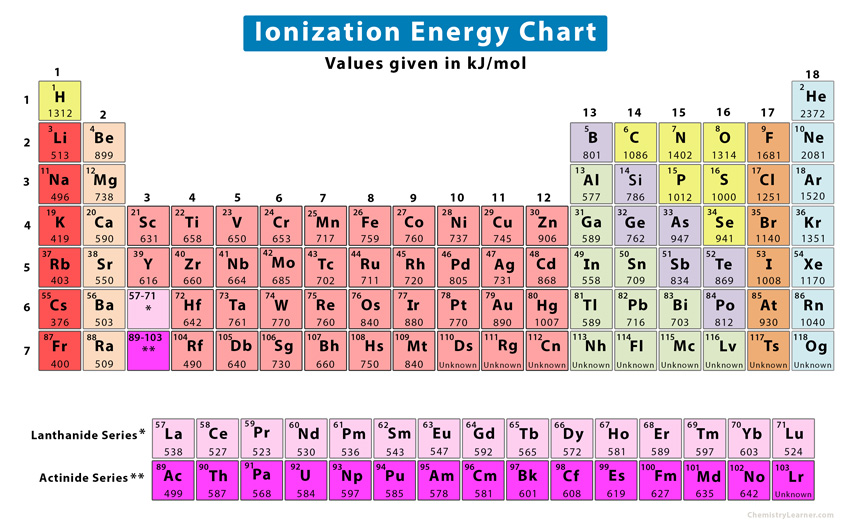
All elements of the periodic table have ionization energies. For some elements, it is easier to remove the electron. They have low ionization energy values. For other elements, it is difficult to remove the electron. These elements have high values.
First Ionization Energy Trend in the Periodic Table
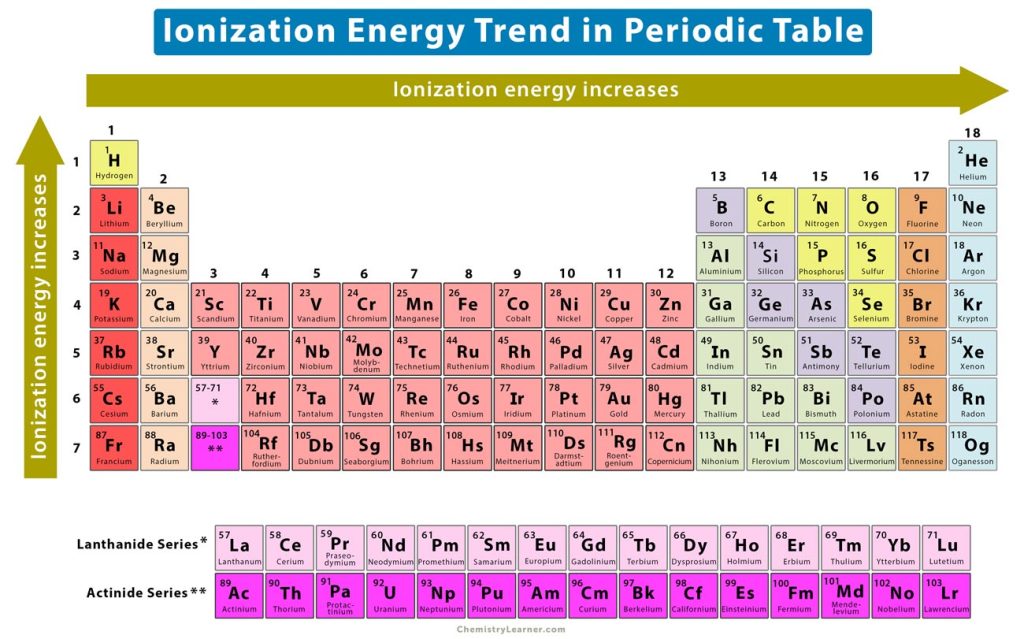
The ionization energy periodic trend is defined as a specific pattern in ionization energy displayed by the elements due to a change in its atomic structure. The trend is observed across a period and down a group in the periodic table [4-8].
1. Horizontal Trend: Ionization Energy Across a Period
From left to right, the atomic number of the elements increases gradually, increasing the number of protons and the nuclear charge. As a result, there will be a stronger attractive electrostatic force between the nucleus and the electrons. This force makes it difficult to remove the electron from the valence shell. Therefore, the ionization energy increases from left to right across a period, as shown in the image above.
Based on this trend, the following conclusions can be made.
- Metals in group 1 have the lowest ionization energies, and noble gases in group 18 have the highest
- Oxygen has lower ionization energy than nitrogen and fluorine
2. Vertical Trend: Ionization Energy Down a Group
From top to bottom, the atomic number increases rapidly. It means that the atoms become larger and increase in size and radius. The valence electrons get further away from the nucleus. Moreover, the inner electrons shield the nucleus from the outer electron, reducing the effective nuclear charge (Zeff). As a result, the valence electrons experience a weak electrostatic attraction and are held loosely to the atom. It makes it easier to remove them from their orbit. Therefore, the ionization energy decreases down a group. In other words, the ionization energy increases from bottom to top, as shown in the image above.
Based on this trend and among the halogens in group 17, fluorine has higher ionization energy than chlorine, bromine, and iodine.
Therefore, the factors affecting ionization energy are atomic size, nuclear charge, and electron shielding.
Exceptions
Exceptions to the above trend exist in the periodic table. The group 2 elements have higher ionization energy than group 13, and group 15 elements have higher values than group 16. The reason is that group 2 elements have completely filled s-subshell, and group 15 elements have half-filled p-subshells. Therefore, these elements require more energy to remove an electron than elements with incompletely filled subshells.
Second, Third, and Higher Ionization Energies
Only the first ionization energy has been discussed so far, in which only one electron is removed from the atom. However, there are higher-order ionization energies [1,3,4].
The amount of energy required to remove an electron from a monovalent cation in its gaseous state is called the second ionization energy. Likewise, the amount of energy required to remove an electron from a divalent cation in its gaseous state is called the third ionization energy, and so on. The different ionization energies are represented by the symbols I1, I2, I3, …, In.
General Chemical Equation for Higher Ionization Energy
Second Ionization Energy (I2): X+ (g) → X2+ (g) + e–
Third Ionization Energy (I3): X2+ (g) → X3+ (g) + e–
And, so on
Why Second Ionization Energy is Higher Than First
The second ionization energy is higher than the first. The reason is that a cation has fewer electrons than its corresponding atom. Hence, the repulsion among the electrons reduces, and the electrons are held tighter by the nucleus. Therefore, the cation will require more energy to ionize further.
Generally, the n-th ionization energy is greater than the (n-1)-th ionization energy, i.e.,
I1 < I2 < I3 … < In
Like the first ionization energy, the second ionization energy is affected by the atomic size, nuclear charge, and electron shielding.
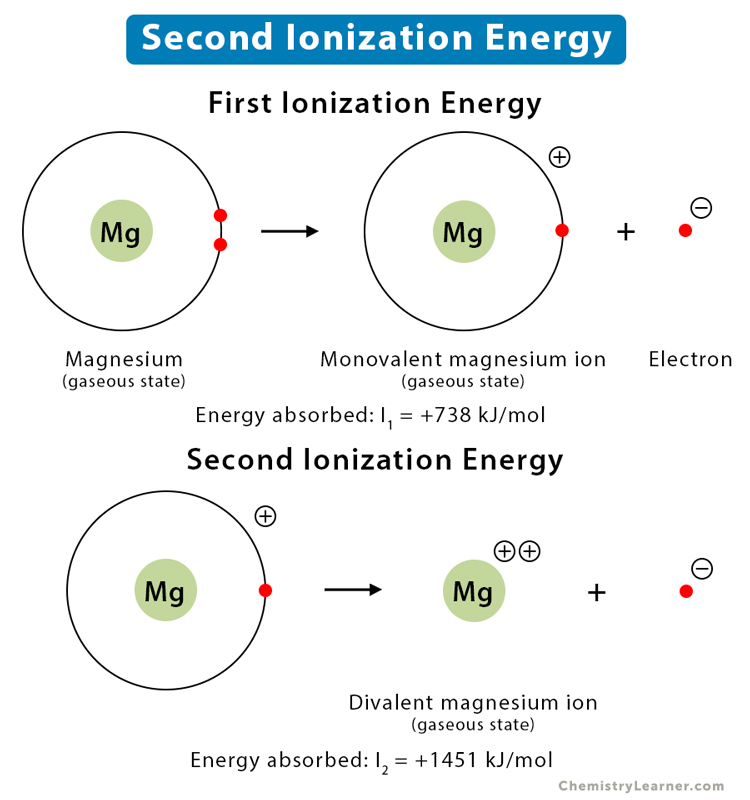
Example
Consider the ionization of the magnesium (Mg) atom, which has a valency of 2. Removing the first electron requires +738 kJ/mol of energy, and removing the second atom requires +1451 kJ/mol amount of energy. It means that the second ionization energy is twice that of the first. The chemical equations are given as follows.
Mg (g) + energy → Mg+ (g) + e– (I1 = +738 kJ/mol)
Mg+ (g) + energy → Mg2+ (g) + e– (I2 = +1451 kJ/mol)
FAQs
Ans. Helium has the largest first ionization energy with a value of 2372 kJ/mol. The reason is that helium is a small atom with a complete 1s-orbital. Due to its stable electron configuration, a large amount of energy is required to remove an electron.
Ans. Francium has the smallest first ionization energy with a value of 400 kJ/mol
Ans. Lithium has the highest second ionization energy because Li+ ion has a stable electron configuration, the same as that of helium. It requires a large amount of energy to remove the electron from the 1s-subshell.
Ans. No. Work function is generally the same as ionization energy. It is associated with the free electrons in a metal.