Titanium Isopropoxide
It is an alkoxide of titanium (IV) and appears as a liquid that is colorless to light yellow in form. Its odor is similar to Isopropyl Alcohol.
Titanium Isopropoxide Synonyms
This substance is also known by many other names like
- Tetraisopropyl Orthotitanate
- TetraIsopropyl Titanate (IV)
- TI Isopropylate
- Tetraisopropoxide Titanium
- Isopropyl Orthotitanate
- Tetraisopropoxytitanium
- Tetraisopropoxyl Orthotitanate
- Titanium Isopropylate
- Tilcom TIPT
- Tyzor TPT
- A1 (Titanate)
- Orgatix TA 10
Titanium Isopropoxide Chemical Formula
The chemical formula for this alkoxide is C12H28O4Ti.
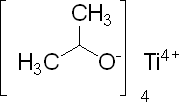
Picture 1 – Titanium Isopropoxide
Source – sigmaaldrich.com
Titanium isopropoxide Uses
This alkoxide is used as laboratory chemical in the manufacture of substances and organic synthesis.
Titanium Isopropoxide Structure
It has a diamagnetic tetrahedral molecule.
CAS Number : 546-68-9
H-Bond Donor : 0
H-Bond Acceptor is 4
Reactions in Air and Water
It is a powerful reducing agent and water-and-air-reactive. It is highly flammable and fumes in air. Its vapors are heavier than air. It is soluble in water. It decomposes quickly in water forming inflammable Isopropyl Alcohol.
Strong Reducing Agent
It is a strong reducing agent and reacts quickly and dangerously with oxygen and other oxidizing agents.
Titanium Isopropoxide Properties
Some of the main physical and chemical properties of this alkoxide are as follows:
- Melting Point : 18 to 20 °C
- Boiling Point : 232 °C.
- Density : 0.962 (g cm-3).
- Molecular Weight : 284.21532 [g/mol]
- Exact Mass : 284.146706
- Flash Point : 66°F
- Topological Polar Surface Area : 92.2
- Heavy Atom Count : 17
- Covalently-Bonded Unit Count : 5
Titanium Isopropoxide MSDS
The Material Safety Data Sheet (MSDS) for this alkoxide is as follows:
Safety Advice for Users
Users need to avoid inhaling it in dust, mist, gas, vapours, or spray form. Contact with this material or inhalation may cause irritation or burn skin or eyes. Users need to wear safety goggles and work under adequate ventilation.
Fire Hazard
This substance can easily be ignited by heat, flames, or sparks. Explosive mixtures may be generated by its vapors with air. There can be a flash back of its vapors after travelling to source of ignition. As most vapors are heavier than air, they will disperse and collect in ground areas (such as sewers and basements). They risk explosion in ground. While in the process of settling in ground, there are chances of fire or explosion.
Fire Safety Precautions
Spray water in large quantities. For spraying, alcohol foam, dry chemical, and carbon dioxide can also be used. All affected containers must be cooled with large quantity of water. Water should be sprayed from as large a distance as possible. It is also important not to allow sprayed water to settle in sewages and other water sources. Soak bulk liquid with cement powder, ash, or other commercial sorbents. Settled materials should be eliminated using suction hoses.
Storage Advice
Dig a holding area (pond, lagoon etc.) to store this substance in liquid or solid form. Soil, sand bags, or foamed concrete may be used to control surface flow. For controlling water spill, natural barricades or spill control booms should be put in place. Other sources of ignition should be kept away from this substance. It should be stored away from water sources.
First Aid
Irritating and/or toxic gases may be produced during fire. Exposure from its vapors may cause dizziness or suffocation. Victims should be shifted to a place of fresh air. Skin and eyes should be flushed with large quantity of running water for at least 15 minutes. If victims are found to be wearing contact lenses in case of a mishap, they should immediately be taken off if found easy to be removed. A doctor should be called to deal with after-effects of exposure to this substance. Artificial respiration should be given in case a sufferer is not found to be breathing. Oxygen should be provided in case a victim finds it difficult to breath. Contaminated clothes should be removed.
- References
- Srlchem.com
- http://cameochemicals.noaa.gov/chemical/22937
- http://pubchem.ncbi.nlm.nih.gov/help.html#xMW
- http://www.chemexper.com/chemicals/supplier/cas/546-68-9.html




