Beryllium Chloride
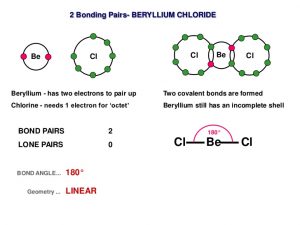
Beryllium chloride, also known as beryllium dichloride in IUPAC nomenclature, is an inorganic compound of beryllium and chloride represented by the chemical formula BeCl2 [1]. It can attract and hold water from the surrounding environment (hygroscopic compound) and dissolves well in several polar solvents [2].
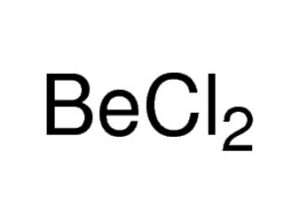
Beryllium Chloride Formula
How is Beryllium Chloride Prepared
It is synthesized by the reaction of beryllium with chlorine at a temperature of about 250 °C, which is represented by the following equation [4]:
Be + Cl2 → BeCl2
When beryllium oxide reacts with chlorine in the presence of carbon at a temperature of about 700-900 °C, it undergoes carbothermal reduction to yield carbon monoxide and beryllium chloride [5]:
BeO + Cl2 + C → BeCl2 + CO
BeCl2 can also be produced by the reaction of beryllium with a diluted solution of hydrogen chloride:
Be + 2HCl → BeCl2 + H2
Reactions with Other Compounds
Beryllium Chloride with Silver Nitrate
Beryllium chloride reacts with silver nitrate dissolved in water, to form silver chloride powder and an aqueous solution of beryllium nitrate:
BeCl2 + 2AgNO3 → Be(NO3)2 + 2AgCl
Properties and Characteristics of Beryllium Chloride
General Properties |
|
| Molar Mass/Molecular Weight | 79.912 g/mol [1] |
Physical Properties |
|
| Color and Appearance | Colorless/white, whitish-yellow, green crystalline mass [1] |
| Odor | Sharp, pungent [1] |
| Melting Point | 415 °C, 779 °F; 399 °C, 750 °F [1] |
| Boiling Point | 482 °C, 899.6 °F [1] |
| Density | 1.9 g cm-3 [1] |
| State of matter at room temperature | Solid [1] |
| Solubility | Soluble in ethyl ether, ethanol, pyridine, and carbon disulfide, insoluble in toluene, benzene [1] |
| Solubility in Water | 71.5 g/100 g at 25 °C [1] |
| Heat Capacity (C) | 71.1 J/mol K, 7.808 J/K |
Atomic Properties |
|
| Crystal Structure | Hexagonal |
What is Beryllium Chloride Used for
- It is a catalyst as well as a chemical intermediate for several beryllium compounds [5].
- Used as a raw material used during the electrolytic extraction of beryllium [6].
Is It Safe
Beryllium chloride causes acute toxicity if inhaled, swallowed, or exposed through the skin. It results in skin corrosion, allergic skin reaction, eye and respiratory tract irritation [1]. It is known to be a carcinogen in humans and may cause organ toxicity, including lung cancer through repeated exposure [1].
- References
- Beryllium Chloride (BeCl2) – Ebi.ac.uk
- Beryllium Chloride – Chem.libretexts.org
- Beryllium Chloride – Chemspider.com
- Beryllium React with Chlorine – Chemiday.com
- Beryllium Chloride – Hazmap.nlm.nih.gov
- Electrolytic Extraction of Beryllium – Tandfonline.com