Bond Order
What is Bond Order
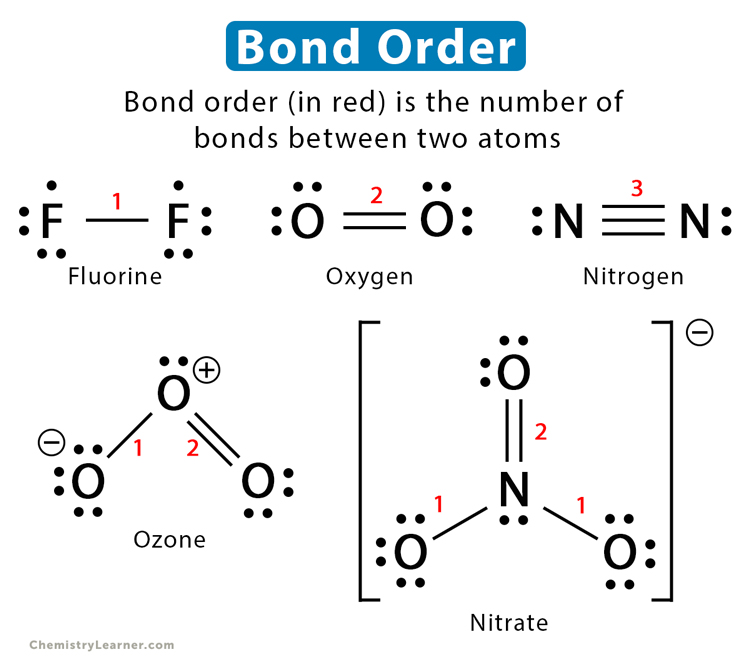
Bond order refers to the number of chemical bonds formed between two atoms in a molecule. For example, two atoms of hydrogen combine to form a hydrogen molecule (H-H or H2). Therefore, the bond order of H2 is one. Similarly, the bond order of oxygen (O=O or O2) is two and that of nitrogen (N≡N or N2) is three. The value need not be an integer, especially for polyatomic molecules having resonance structures [1-4].
The bond order indicates the stability of the bond. It is inversely proportional to the bond length. The shorter the bond length, the higher is the bond order.
American chemist Linus Pauling introduced bond order.
How to Find Bond Order
1. Using Lewis Structure
The bond order can be determined from the Lewis structure using the following steps [1,2,5,6]:
Step 1: Draw the Lewis structure
Step 2: Allocate the following bond order for different types of bonds:
- 0: No bond
- 1: Single covalent bond
- 2: Double covalent bond
- 3: Triple covalent bond
Step 3: Count the total number of bonded pairs of atoms or bond group
Step 4: Add the bond order of each bond group
Step 5: Divide the total bond order by the number of bond groups to get an average bond order
Examples
1: Nitrate ion (NO3–)
The number of bonds in different bond groups is as follows.
N-O: 1
N-O: 1
N=O: 2
Total bond order: 1 + 1 + 2 = 4
Number of bond groups: 3
The bond order of the molecule is: 4/3 = 1.33
2. Ethylene (C2H2 or H-C≡C-H)
The number of bonds in different bond groups is as follows.
C-H: 1
C-H: 1
C≡C: 3
Total bond order: 1 + 1 +3 = 5
Total number of bond groups: 3
The bond order of the molecule is: 5/3 = 1.67
3. Sulfur dioxide (SO2 or O-S=O)
The number of bonds in different bond groups is as follows.
O-S: 1
S=O: 2
Total bond order: 1 + 2 = 3
Total number of bond groups: 2
The bond order of the molecule is: 3/2 = 1.5
4. Ozone (O3 or O-O=O)
The number of bonds in different bond groups is as follows.
O-O: 1
O=O: 2
Total bond order: 1 + 2 = 3
Total number of bond groups: 2
The bond order of the molecule is: 3/2 = 1.5
Sulfur dioxide and ozone are isoelectronic and have the same bond order. All isoelectronic species have the same bond order.
2. Using Molecular Orbital Theory
According to molecular orbital theory, the bond order is the difference between the number of bonding and anti-bonding electrons divided by two. The formula is given by [7]:
Bond order = (Number of bonding electrons – number of anti-bonding electrons)/2
Examples
1. Carbon monoxide (CO)
The molecular configuration of CO is (σ1s)2(σ*1s)2(σ2s)2(σ*2s)2(π)4(2pz)2.
Number of bonding electrons = 10
Number of anti-bonding electrons = 4
Bond order = (10-4)/2 = 3
2. Nitrosonium (NO+)
The molecular configuration of NO+ is (σ1s)2(σ*1s)2(σ2s)2(σ*2s)2(π2px)2(π2py)2(σ2pz)2(π*2px)1.
Number of bonding electrons = 10
Number of anti-bonding electrons = 5
Bond order = (10-5)/2 = 2.5
The following table gives the bond order of a few chemical species [8]:
| Species (molecule or ion) | Total number of bonding electrons | Total number of anti-bonding electrons | Bond order |
| Hydrogen (H2) | 2 | 0 | 1 |
| Oxygen (O2) | 10 | 6 | 2 |
| Fluorine (F2) | 10 | 8 | 1 |
| Dicarbon (C2) | 8 | 4 | 2 |
| Diboron (B2) | 6 | 4 | 1 |
| Dilithium (Li2) | 2 | 0 | 1 |
| Diberyllium (Be2) | 4 | 4 | 0 |
| Cyanide (CN–) | 10 | 4 | 3 |