Carboxylic Acid
Carboxylic acids are a fundamental and versatile group of organic compounds that play a pivotal role in natural and synthetic chemistry. They are characterized by the carboxyl functional group (-COOH), a combination of a carbonyl group (-C=O) and a hydroxyl group (-OH) bonded to the same carbon atom. This unique structural feature imparts distinctive properties and reactivity, making carboxylic acids continue to fascinate chemists. [1-4]
Structure
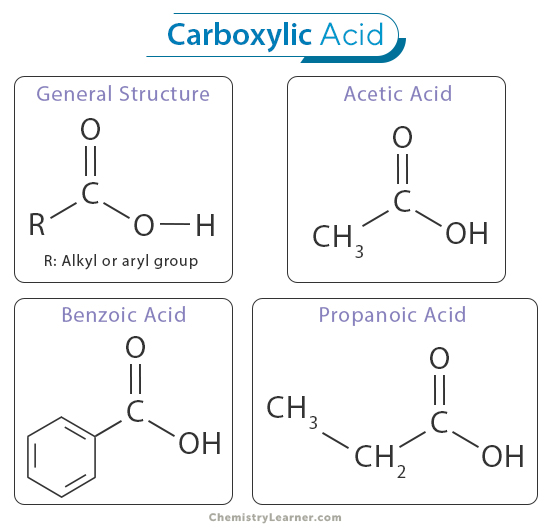
The structure of carboxylic acids is a critical element that sets them apart from other functional groups in organic chemistry. Carboxylic acids consist of a carboxyl active group (-COOH) responsible for their distinctive properties and behavior. This functional group comprises a carbonyl group (C=O), where a carbon atom is double-bonded to an oxygen atom, and a hydroxyl group (OH), where an oxygen atom is bonded to a hydrogen atom, both of which are attached to the same carbon atom. [1-6]
Nomenclature
The general IUPAC naming convention for carboxylic acids involves three main components: the parent alkane chain, the carboxyl group, and any substituent groups. The parent alkane chain is selected based on the longest continuous carbon chain, including carboxyl carbon. The suffix “-oic acid” is then added to indicate the presence of the carboxyl group. For example, the carboxylic acid with a three-carbon parent chain is named “propanoic acid.” Another example is that 2-methylbutanoic acid denotes a carboxylic acid with a four-carbon parent chain (butanoic acid) and a methyl group (CH3) attached to the second carbon atom. [1-6]
Carboxylic acids can also have common names, often derived from their historical or commonly used names. For instance, acetic acid is a common name for ethanoic acid, which contains a two-carbon parent chain.
Is Carboxylic Acid Polar
Carboxylic acids, with their distinctive molecular structure, are indeed polar compounds. The polarity of carboxylic acids arises from the highly electronegative oxygen atoms within the carboxyl functional group. The oxygen atom in the carbonyl group draws electron density away from the carbon atom, creating a partially positive charge on the carbon and a partially negative charge on the oxygen. Similarly, the oxygen atom in the hydroxyl group is more electronegative than the hydrogen atom, contributing to a partial negative charge on the oxygen and a partial positive charge on the hydrogen. This inherent polarity makes carboxylic acids capable of forming hydrogen bonds and interacting strongly with other polar molecules. Also, this polarity makes the carbon atom electrophilic, rendering it susceptible to nucleophilic attacks. [1,2]
Is Carboxylic Acid a Strong Acid
Carboxylic acids are often considered moderate to weak acids rather than strong. Unlike strong acids such as hydrochloric or sulfuric acid, carboxylic acids do not readily dissociate into hydrogen ions and anions when dissolved in water. As a result, their dissociation is partial, and they maintain some of their original molecular form in solution. [1,2]
RCOOH + H2O ⇌ RCOO– + H3O+
Properties
We have already discussed the polarity and acidity of carboxylic acids. Here are some other properties: [1,2,4]
Hydrogen Bonding: Carboxylic acids are excellent hydrogen bond donors and acceptors due to the presence of the hydroxyl group. This property leads to the formation of strong intermolecular forces in carboxylic acid compounds.
Melting and Boiling Points: Carboxylic acids generally have higher melting and boiling points than other organic compounds of similar molecular weight. It is due to the presence of hydrogen bonds between the molecules.
Solubility: Carboxylic acids are typically soluble in water. Their ability to form hydrogen bonds with water molecules allows them to dissolve readily in aqueous solutions.
Odor: Many carboxylic acids have distinctive and often pungent odors. For example, acetic acid (vinegar) and butyric acid (found in rancid butter) are known for their characteristic smells.
Molecular Weight: The molecular weight of carboxylic acids can vary widely, from simple compounds like formic acid (HCOOH) to larger, more complex molecules with multiple carboxyl groups.
Biological Significance: Carboxylic acids play crucial roles in biological systems, as they are essential components of amino acids, fatty acids, and the citric acid cycle, a central metabolic pathway.
Synthesis
Carboxylic acids can be synthesized from various starting materials, and the choice of method often depends on the specific target compound and the desired reaction conditions. Here, we explore some principal techniques. [1,2]
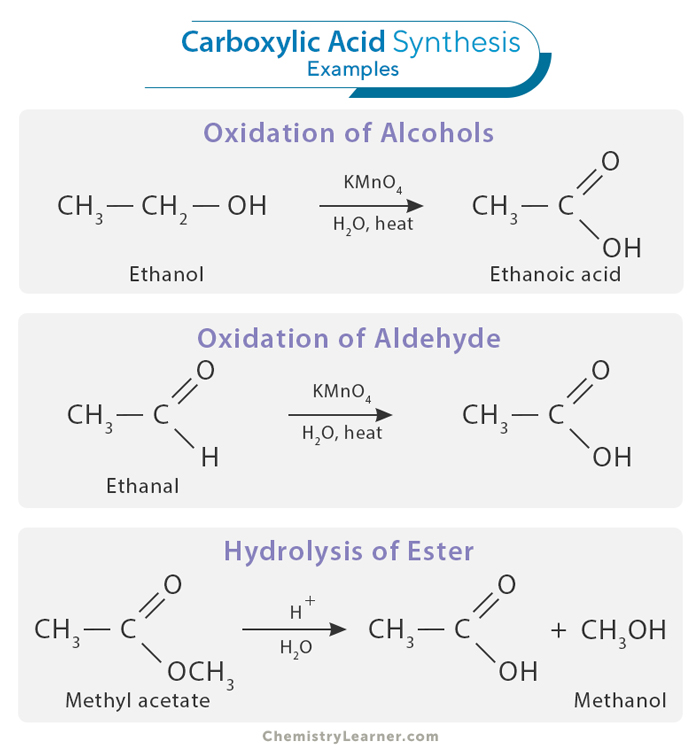
1. Oxidation of Aldehydes and Primary Alcohols
One of the most widely used methods for synthesizing carboxylic acids is the oxidation of aldehydes and primary alcohols. This process can be carried out using an oxidizing agent like sodium or potassium dichromate in dilute sulfuric acid. When an aldehyde is oxidized, it is converted into the corresponding carboxylic acid. Similarly, primary alcohols are oxidized to carboxylic acids by forming an aldehyde intermediate.
2. Hydrolysis of Esters
Esters, which contain the carbonyl group (C=O) and an alkoxy group (OR) bonded to the same carbon atom, can be hydrolyzed to produce carboxylic acids. An acid or a base typically catalyzes this hydrolysis reaction. Acid-catalyzed hydrolysis of esters involves using a strong hydrochloric acid to break the ester bond and release the carboxylic acid and alcohol. Base-catalyzed hydrolysis employs a strong base, like sodium hydroxide, to produce a carboxylate ion.
Reactions of Carboxylic Acids
Carboxylic acids are highly versatile compounds that are involved in various chemical reactions. These reactions are fundamental to the synthesis of various organic compounds, the preparation of pharmaceuticals, and the operation of biological processes. Here, we explore some of the most important reactions of carboxylic acids, along with specific examples illustrating their significance. [1,2,5,6]
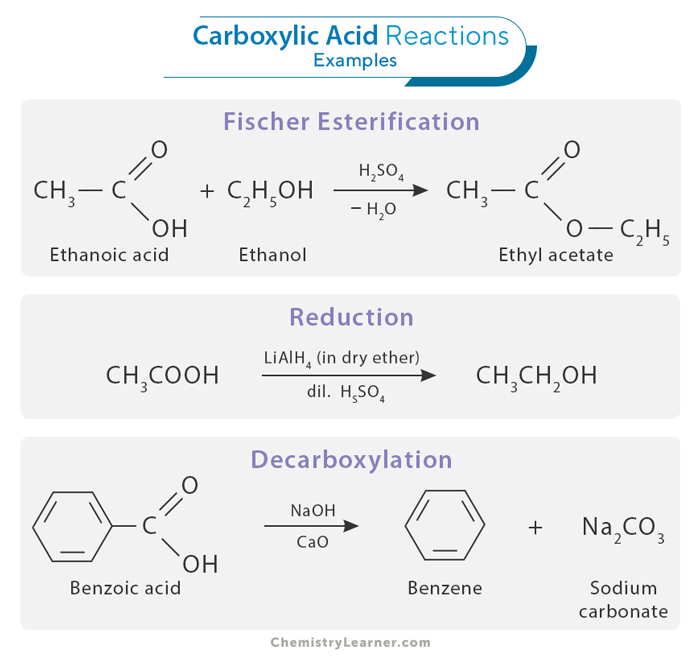
1. Esterification
Fischer Esterification is a common reaction involving carboxylic acids. It occurs when a carboxylic acid reacts with an alcohol in the presence of an acid catalyst to form an ester and water. For example, acetic acid can react with ethanol (C2H5OH) in the presence of a sulfuric acid (H2SO4) catalyst to produce ethyl acetate (CH3COOC2H5) and water.
CH3COOH + C2H5OH → CH3COC2H5 + H2O
2. Reduction
Carboxylic acids can be reduced to alcohols through various reduction methods. One common approach is reducing agents like lithium aluminum hydride (LiAlH4) or sodium borohydride (NaBH4). For example, propanoic acid (CH3CH2COOH) can be reduced to form propanol (CH3CH2CH2OH) using a reducing agent.
CH3CH2COOH + LiAlH4 → CH3CH2CH2OH
3. Decarboxylation
Decarboxylation is a reaction in which a carboxylic acid loses a carbon dioxide molecule, forming an alkane. This reaction is often carried out at elevated temperatures. For example, decarboxylation of acetic acid (CH3COOH) produces methane (CH4) and carbon dioxide (CO2).
CH3COOH → CH4 + CO2
Uses
Here are some essential uses of carboxylic acids: [7]
- Soap production requires higher fatty acids since soaps are sodium or potassium salts derived from higher fatty acids like stearic acid.
- In the food industry, various organic acids are employed in manufacturing beverages and food products. Acetic acid is utilized in vinegar production. Sodium salts of organic acids are used as preservatives.
- Within the pharmaceutical sector, organic acids play a significant role in the composition of various drugs, such as aspirin and phenacetin.
- Acetic acids are commonly utilized as a coagulant in the rubber manufacturing process.
- Organic acids have extensive applications in producing dyestuffs, perfumes, and rayon.





Solve an example
If you mean solve a problem, we do not have any problem for this article.
I want always to teach me senior three work cause I loved ur explanations