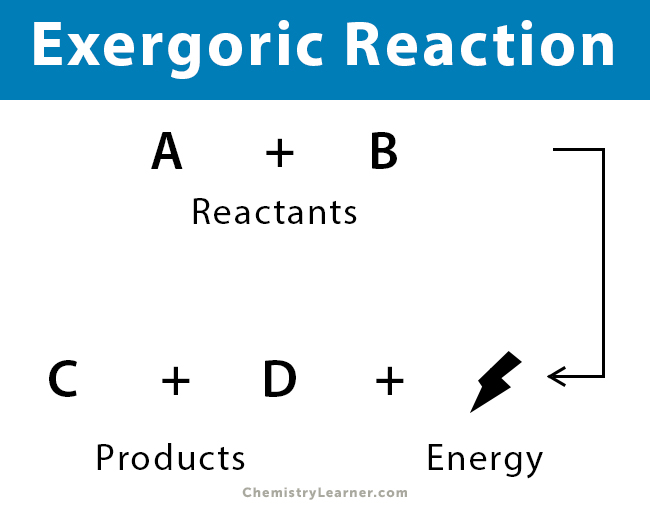Exergonic Reaction
What is an Exergonic Reaction
An exergonic reaction is a chemical reaction in which the reaction (system) gives out energy to the surroundings. It is the opposite of an endergonic reaction. In this process, old bonds are broken, and new bonds are formed, thereby releasing energy. The change in Gibbs’ free energy (ΔG) is negative, i.e., the reactants have more free energy than the products. Exergonic reactions are spontaneous reactions since they can occur without adding external energy to the system. All exothermic reactions are exergonic, but not all exergonic reactions are exothermic [1-5].
General Equation
Reactants → Products + Energy
What Happens to Energy in an Exergonic Reaction
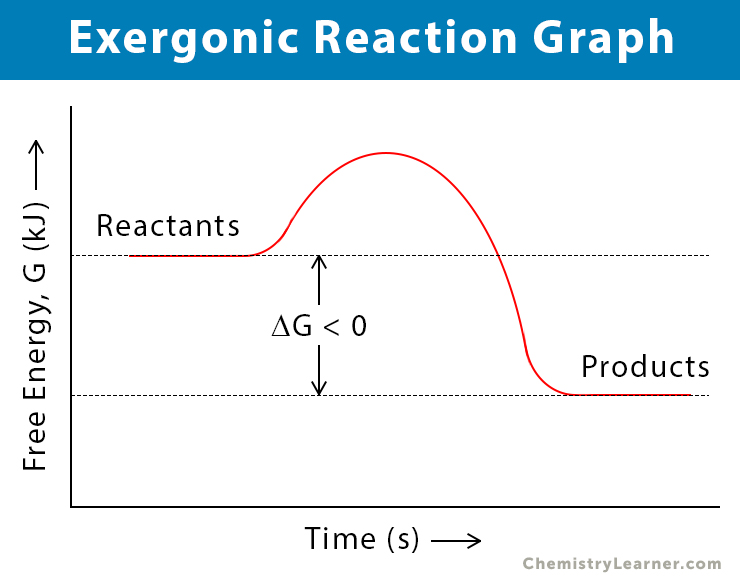
The energy required to initiate an exergonic reaction is called activation energy. After overcoming this activation barrier, the reactants react and form products. The Gibbs’ free energy of the reactants (GR) is higher than that of the products (GP). Therefore, the change in free energy is negative [6].
ΔG = GP – GR < 0
Where,
GR: Gibbs’ free energy of the reactants
GP: Gibbs’ free energy of the products
Exergonic Reaction Examples
Here are some examples of the exergonic reaction.
1. Mixing of sodium (Na) and chlorine (Cl) to form sodium chloride (NaCl)
2 Na + Cl2 → 2 NaCl + energy
2. Hydrolysis of adenosine triphosphate (ATP) to adenosine diphosphate (ATD)
ATP + H2O → ADP + Pi + energy
3. Combustion of Propane (C3H8) with Oxygen (O2)
5 O2 + C3H8 → 4 H2O + 3 CO2 + energy
Endergonic and Exergonic Reactions
Exergonic reactions can be coupled to endergonic reactions. An example of such coupling is the oxidation-reduction (redox) reaction. Both reactions require energy to proceed. They use enzymes to lower the activation energy needed to complete the reaction. Enzymes often act by coupling an endergonic reaction to the exergonic reaction during the hydrolysis of ATP [7].
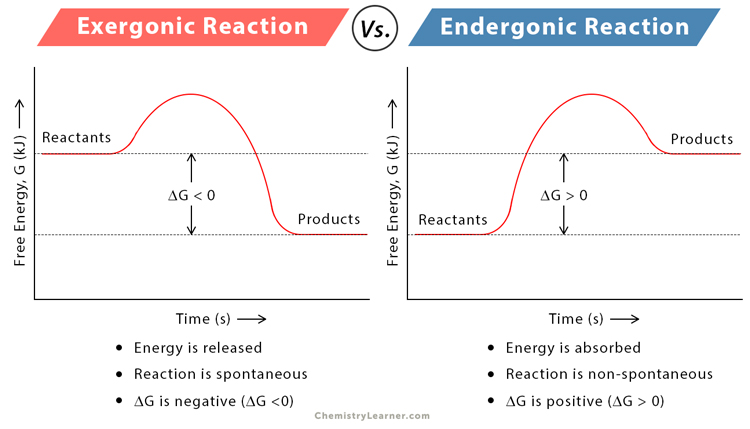
Exergonic vs. Endergonic Reactions
The following table list the basic differences between exergonic and endergonic reactions [8].
| Exergonic Reaction | Endergonic Reaction | |
|---|---|---|
| Definition | Gives off energy | Absorbs energy |
| Input energy | Not required | Required |
| Energy of reactants | Higher than the products | Lower than the products |
| Change in Gibbs’ free energy (ΔG) | Negative | Positive |
| Entropy | Increased | Decreased |
| Spontaneity | Favorable and spontaneous | Less favorable and non-spontaneous |
| Exothermic and endothermic reactions | Exothermic reactions are exergonic | Endothermic reactions are endergonic |
| At normal conditions | Occurs | Does not occur |