Electrophilic Aromatic Substitution
- Definition: What is Electrophilic Aromatic Substitution?
- Types of Electrophilic Aromatic Substitution
- Examples of Electrophilic Aromatic Substitution of Benzene
- Examples of Electrophilic Aromatic Substitution of Other Aromatic Compounds
- Mechanism of Electrophilic Aromatic Substitution of Benzene [9-14]
- FAQs
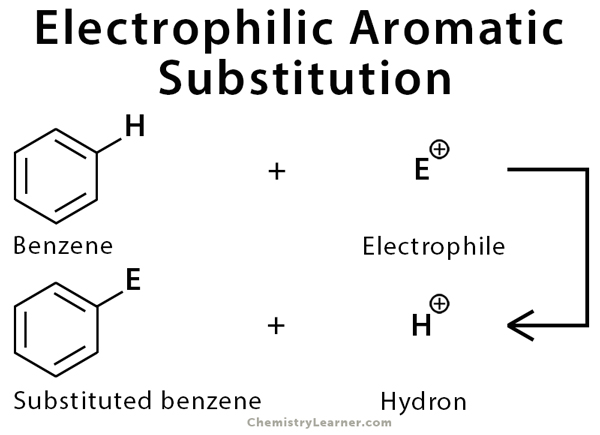
Definition: What is Electrophilic Aromatic Substitution?
Electrophilic aromatic substitution is a chemical reaction in which a hydrogen atom or a functional group from an aromatic compound is substituted with another functional group, which is an electrophile [1-3].
Types of Electrophilic Aromatic Substitution
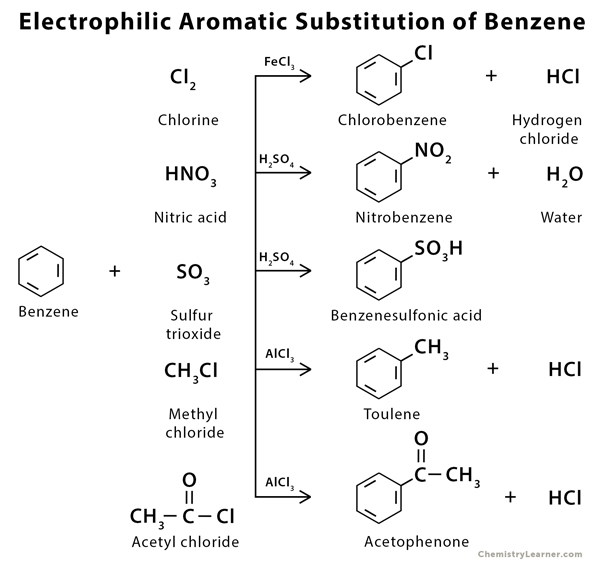
There are five different types of electrophilic aromatic substitution [2].
- Halogenation – replacing with chlorine (Cl) (chlorination) or bromine (Br) (bromination)
- Nitration – replacing with a nitrate group (NO2)
- Sulfonation – replacing with bisulfite (SO3H)
- Friedel-Crafts Alkylation – replacing with an alkyl group (R)
- Friedel-Crafts Acylation – replacing with an acyl group (RCO)
Examples of Electrophilic Aromatic Substitution of Benzene
Benzene can undergo electrophilic substitution reaction giving a wide range of compounds [2].
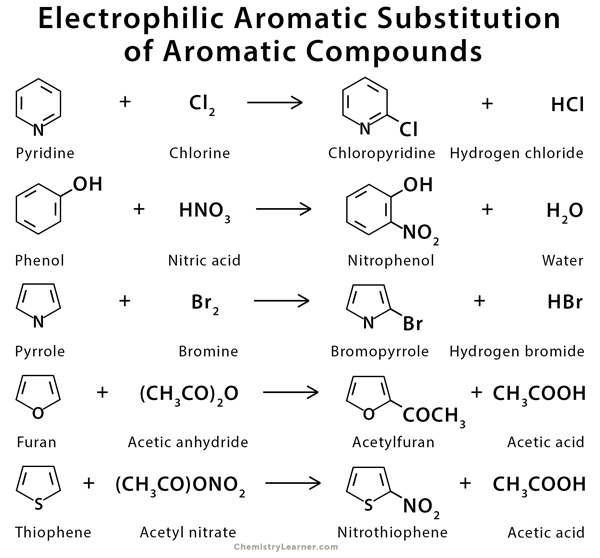
Examples of Electrophilic Aromatic Substitution of Other Aromatic Compounds
Aromatic compounds like phenol, aniline, naphthalene, pyrrole, furan, thiophene, pyridine, indole, and imidazole can undergo electrophilic substitution reaction. Their mechanism is similar to benzene [4-8].
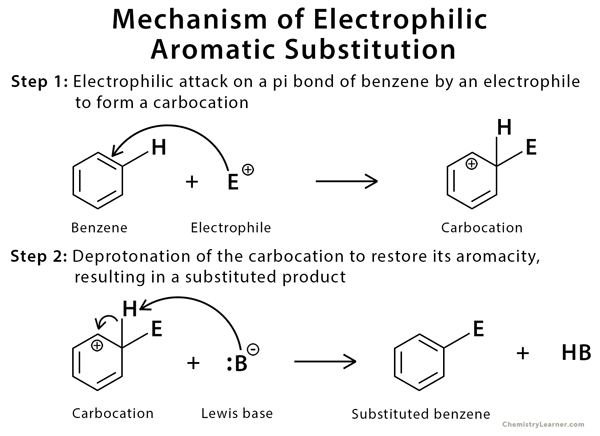
Mechanism of Electrophilic Aromatic Substitution of Benzene [9-14]
FAQs
Ans. Aromatic compounds like benzene undergo substitution reaction rather than addition reactions because addition reactions disrupt the delocalization of the pi molecular orbital, reducing the stability of the benzene ring. In contrast, substitution reactions only cause temporary disruption, thereby allowing the compound to retain its aromaticity.
Ans. Phenol has a higher electron density than benzene, thereby making it easier for the electrophile to attack.
Ans. Oxygen, being more electronegative than nitrogen, distributes more negative charge density upon itself and less upon the ring, thus stabilizing the carbocation intermediate less, making furan less reactive towards EAS than pyrrole.
References
- Definition – Study.com
- Definition – Masterorganicchemistry.com
- Definition – Byjus.com
- Example – Chemistry.msu.edu
- Example – Byjus.com
- Example – Onlineorganicchemistrytutor.com
- Example – Chem.gla.ac.uk
- Example –Webpages.iust.ac.ir
- Mechanism – Masterorganicchemistry.com
- Mechanism – Byjus.com
- Mechanism – Cliffsnotes.com
- Mechanism – Chemguide.co.uk
- Mechanism – Chem.ucla.edu
- Mechanism – Chem.ucalgary.ca





It is the best way of expressing electrophilic reactions. Please continue in other areas of chemistry.