Hassium
What is Hassium
Hassium (pronunciation: HAS-ee-em) [1] is a synthetic [2], highly radioactive chemical element [1, 3] belonging to the transition metal family, represented by the chemical symbol Hs [2]. It is thought to have a solid metallic, silvery look and is quite a difficult element to study because only a few atoms have been made to date [1].
It has 15 isotopes [3], out of which the most stable one is hassium-276, with a half-life period of 1.1 hours [3]. As of now, the final product obtained after the radioactive decay of this isotope is not known [5].
History
Origin of its Name: The element is named after Hesse, the German state where it was first created [3].
Who Discovered Hassium: The German physicists Peter Armbruster and Gottfried Münzenberg are credited with discovering this element [3].
How was Hassium Discovered
In 1978, a research team headed by two Russian physicists Vladimir Utyonkov and Yuri Oganessian made the first effort to create element 108 at the JINR (Joint Institute for Nuclear Research) in Russia [3]. They discovered the new isotope 270 by bombarding radium with calcium [3]. Five years later, they bombarded bismuth with manganese to synthesize isotope 263, bombarded lead with iron to create isotope 264, and californium with neon to get isotope 270 [3].
In 1984, a team of researchers headed by Gottfried Münzenberg and Peter Armbruster created isotope 265 at GSI (Gesellschaft für Schwerionenforschung) in Germany by bombarding lead with iron [3]. The data from GSI was much more reliable and accurate as compared to that from JINR, and thus, the team from GSI was given the rights to name the element [3].
Classification and Position of Hassium on the Periodic Table
| Group | 8 [3] |
| Period | 7 [3] |
| Block | d [3] |
Properties and Characteristics of Hassium
General Properties |
|||||||||||
| Relative atomic mass | 269 [3] | ||||||||||
| CAS number | 54037-57-9 [3] | ||||||||||
Physical Properties |
|||||||||||
| Color/appearance | Presumably silvery white/ gray [6] | ||||||||||
| Melting point/freezing point | Unknown [3] | ||||||||||
| Boiling point | Unknown [3] | ||||||||||
| Density | 41 kg cm-3 (estimated) [6] | ||||||||||
| State of matter at room temperature (solid/liquid/gas) | Solid [3] | ||||||||||
| Hardness (Brinell, Mohs, Vickers) | Unknown [7] | ||||||||||
| Electrical conductivity | Unknown [7] | ||||||||||
| Thermal (heat) conductivity | Unknown [7] | ||||||||||
| Specific heat | Unknown [3, 7] | ||||||||||
| Bulk modulus | Unknown [3] | ||||||||||
| Shear modulus | Unknown [3] | ||||||||||
| Young’s modulus | Unknown [3] | ||||||||||
| Vapor pressure | |||||||||||
| Temperature (K) | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 | 2200 | 2400 |
| Pressure (Pa) | – | – | – | – | – | – | – | – | – | – | – |
Chemical Properties |
|||||||||||
| Flammability | Unknown | ||||||||||
| Oxidation state/Oxidation number | [+8] [3] | ||||||||||
Atomic Data of Hassium
| Atomic number | 108 [1, 3] | |||||||
| Valence electrons | 2 | |||||||
| Quantum numbers | ||||||||
| – n | 6 [8] | |||||||
| – ℓ | 2 [8] | |||||||
| – mℓ | -2 [8] | |||||||
| – ms | -1/2 [8] | |||||||
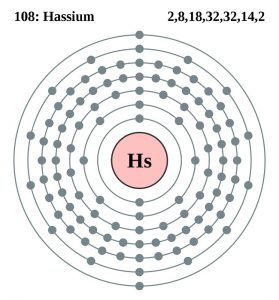
| Electron configuration (noble gas configuration) | [Rn] 5f146d67s2 [3] | |||||||
| Atomic structure | ||||||||
| – Number of electrons | 108 [6] | |||||||
| – Number of neutrons | 157 [6] | |||||||
| – Number of protons | 108 [6] | |||||||
| Radius of Atom | ||||||||
| – Atomic radius (Å) | Unknown [3] | |||||||
| – Covalent radius | 1.34 Å [3] | |||||||
| Electronegativity (Pauling-scale) | Unknown [3] | |||||||
| Electron affinity (kJ mol-1) | Unknown [3] | |||||||
| Ionization energy (kJ mol-1) | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
| – | – | – | – | – | – | – | – | |
What is Hassium used for
Since Hassium is not a naturally occurring element and has never been created in abundance, its use is now restricted only to research studies [3]. It is believed that the radioactive element might react with the other elements of its group if produced in large amounts [9].
Interesting Fact about Hassium
- Since Hassium does not have its own image, it has been represented with the state of Hesse’s coat of arms logo to honor the German state [3].
- References
- https://education.jlab.org/itselemental/ele108.html
- http://hobart.k12.in.us/ksms/PeriodicTable/hassium.htm
- http://www.rsc.org/periodic-table/element/108/hassium
- https://education.jlab.org/itselemental/iso108.html
- http://www.periodictable.com/Isotopes/108.276/index.html
- https://www.americanelements.com/hassium.html
- http://periodictable.com/Elements/108/data.html
- http://chemistry-reference.com/q_elements.asp?Symbol=Hs&language=en
- http://www.elementsdatabase.com/Hassium-Hs-108-element/