Americium
What is Americium
Americium (pronunciation: am-er-ISH-ee-em) is a shiny, silvery, synthetic element belonging to the actinide series and is represented by the chemical symbol Am [1, 2, 3]. Since it is artificially created, it does not have any known stable isotopes [4]. However, it has nineteen radioisotopes, out of which the most stable are 243Am and 241Am characterized by a half-life period of 7,370 years and 432.2 years respectively [4].
Where is Americium Found
It is naturally found in trace quantities in uranium mineral ores [1]. The element is commercially produced by bombarding plutonium with neutrons in nuclear reactors [1]. It is also produced during the explosion of nuclear weapons [1].
History
Origin of its Name: The element is named after America, the country where it was discovered [1].
Who discovered it: The American chemist Glenn Theodore Seaborg, as well as his colleagues Ralph James, Albert Ghiorso, and Leon Morgan, are credited for the discovery of americium [1].
When and How was it Discovered
In 1944, Glenn Seaborg along with his team of researchers while working at the University of Chicago’s Wartime Metallurgical Laboratory produced this transuranium element through the bombardment of plutonium-239 by neutrons to produce plutonium-240, which was again bombarded to produce plutonium-241 [1, 3]. The plutonium-241 underwent radioactive decay to form americium-241 [3]. The discovery was announced by Seaborg in late 1945 [3].
Americium Identification |
|||
| Atomic number | 95 [1] | ||
| CAS number | 7440-35-9 [1] | ||
| Position in the periodic table | Group | Period | Block |
| Actinides [1] | 7 [1] | f [1] | |
Properties and Characteristics of Americium
General Properties |
||||||||||||||||
| Relative atomic mass | 243 [1] | |||||||||||||||
| Atomic mass | 243 amu [1] | |||||||||||||||
| Molar mass | 243.061 g/mol [5, 6] | |||||||||||||||
Physical Properties |
||||||||||||||||
| Color | Silvery-white [1, 7] | |||||||||||||||
| Melting point/freezing point | 1176 °C, 2149 °F [1] | |||||||||||||||
| Boiling point | 2011 °C, 3652 °F [1] | |||||||||||||||
| Density | 12 g cm-3 [1] | |||||||||||||||
| State of matter at room temperature (solid/liquid/gas) | Solid [1, 7] | |||||||||||||||
| Hardness | ||||||||||||||||
| – Brinell | Unknown [8] | |||||||||||||||
| – Mohs | Unknown [8] | |||||||||||||||
| – Vickers | Unknown [8] | |||||||||||||||
| Electrical Conductivity | Unknown [8] | |||||||||||||||
| Thermal (heat) conductivity | 10 W/(m K) [8] | |||||||||||||||
| Specific heat | Unknown [1] | |||||||||||||||
| Bulk modulus | Unknown [1] | |||||||||||||||
| Shear modulus | Unknown [1] | |||||||||||||||
| Young’s modulus | Unknown [1] | |||||||||||||||
| Vapor pressure | ||||||||||||||||
| – Temperature (K) | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 | 2200 | 2400 | |||||
| – Pressure (Pa) | – | – | 3.88X 10-7 | 1.67X 10-3 | 0.423 | 21.35 | – | – | – | – | – | |||||
Chemical Properties |
||||||||||||||||
| Oxidation state/Oxidation number | +2, +3, +4, +5, +6, (+7) [1] | |||||||||||||||
| Isotopes | Isotope | Mass | Abundance (%) | Half-life | Mode of decay | |||||||||||
| 241Am | 241.057 | – | 432.7 y | α | ||||||||||||
| 1.2 X 1014 y | sf | |||||||||||||||
| 243Am | 243.061 | – | 7.37 X 103 y | α | ||||||||||||
| 2 X 1014 y | sf | |||||||||||||||
Atomic Data of Americium (Element 95)
| Valence electrons | 2 [9] | |||||||
| Quantum numbers | ||||||||
| – n | 5 [9] | |||||||
| – ℓ | 3 [9] | |||||||
| – mℓ | 3 [9] | |||||||
| – ms | +1/2 [9] | |||||||
| Electron configuration (noble gas configuration) | [Rn] 5f77s2 [1] | |||||||
| Atomic structure | ||||||||
| – Number of electrons | 95 [6] | |||||||
| – Number of neutrons | 148 [6] | |||||||
| – Number of protons | 95 [6] | |||||||
| Radius of Atom | ||||||||
| – Atomic radius | 2.44 Å [1] | |||||||
| – Covalent radius | 1.73 Å [1] | |||||||
| Electronegativity (Pauling-scale) | Unknown [1] | |||||||
| Electron affinity | Unknown [1] | |||||||
| Ionization energy (kJ mol-1) | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
| 576.384 | – | – | – | – | – | – | – | |
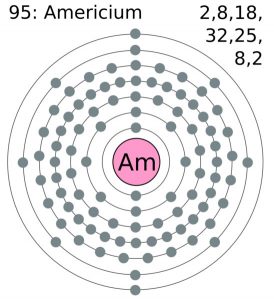
Americium Electron Configuration (Bohr Model)
What is It Used for
- Small amounts of americium dioxide are used in smoke detectors and alarms [1, 6].
- Several isotopes of americium are now investigated on their use as a possible power source for deep space vehicles [1, 3]. It could potentially become the favored fuel source and replace the commonly used plutonium [3].
- As a portable source of alpha and gamma rays, it has plenty of industrial and medical applications like spectroscopy and radiography [3].
Toxicity of Americium
Americium is known to be toxic because of its radioactivity and exposure to this radiation can cause genetic changes in human cells, resulting in bone cancers [10]. High levels of radiation from americium can damage the lungs, liver, thyroid, and kidneys [10].
Interesting Facts
- The element is represented with a smoke alarm-shaped object painted with the colors of the American flag, indicating the place of its origin [1].
- At room temperature, it discolors slowly in dry air [6].
Cost of Americium
The price of pure americium is $1,500 per gram [6].
- References
- http://www.rsc.org/periodic-table/element/95/americium
- https://education.jlab.org/itselemental/ele095.html
- https://www.livescience.com/39874-facts-about-americium.html
- https://education.jlab.org/itselemental/iso095.html
- https://pubchem.ncbi.nlm.nih.gov/compound/105139#section=Top
- https://www.convertunits.com/molarmass/Americium
- https://www.chemicool.com/elements/americium.html
- http://periodictable.com/Elements/095/data.html
- http://chemistry-reference.com/q_elements.asp?language=en&Symbol=Am
- https://www.atsdr.cdc.gov/phs/phs.asp?id=809&tid=158