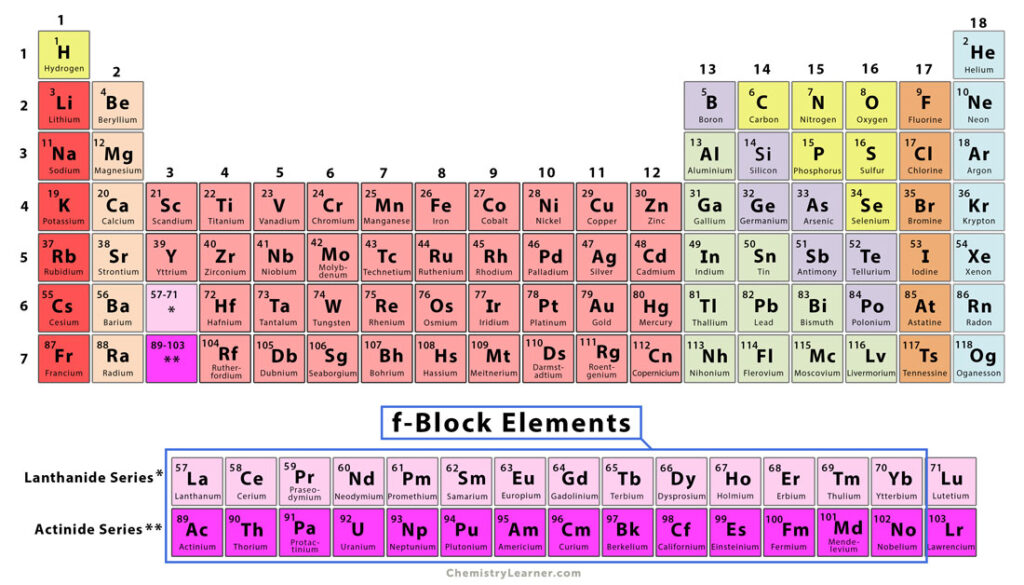
f-Block Elements
The periodic table elements whose last electron goes into the f subshell are known as f-block elements. These elements are usually not considered part of any group. They are sometimes called inner transition metals because they offer a transition in properties between the s-block and d-block elements of Periods 6 and 7. Moreover, they have characteristics absent from many other elements [1-4].
Location
The f-block elements appear in two series, known as lanthanides and actinides. Lanthanides are those elements whose last electron goes to the 4f subshell. Actinides are those elements whose 5f subshell is progressively filled [1-4].
Both lanthanides and actinides a placed separately at the bottom of the periodic table, i.e., below the main body. One reason is that their properties are different from s-block and p-block elements. Another reason is that they fill up too much space and for the simplicity of printing on a standard letter size paper.
Electron Configuration
Generally, electrons are fed into orbitals in order of increasing energy until all the electrons have been accommodated. The electronic configuration of the f-block elements is done by filling electrons in deep-rooted 4f and 5f subshells with increasing atomic numbers. The seven f orbitals can accommodate up to seven pairs of electrons. Hence, the f-block occupies fourteen columns in the periodic table, resulting in 28 f-block elements. The atomic number of lanthanides ranges from 57 to 71, and that of actinides ranges from 89 to 103 [1-4].
The general valence shell electron configuration of f-block elements is (n − 2)f0, 2 to 14 (n − 1)d0 to 2 ns2. The general electron configuration of lanthanides is [Xe] 4f0, 2 to 14 5d0 to 2 6s2, and that of actinides is [Rn] 5f0, 2 to 14 6d0 to 2 7s2. A list of all f-block elements, their chemical symbols, atomic numbers, and valence shell electron configuration is given in the table below.
| Lanthanide | Chemical Symbol | Atomic Number | Valence Shell Electron Configuration | Actinide | Chemical Symbol | Atomic Number | Valence Shell Electron Configuration |
|---|---|---|---|---|---|---|---|
| Lanthanum | La | 57 | 5d1 6s2 | Actinium | Ac | 89 | 6d1 7s2 |
| Cerium | Ce | 58 | 4f1 5d1 6s2 | Thorium | Th | 90 | 6d2 7s2 |
| Praseodymium | Pr | 59 | 4f3 6s2 | Protactinium | Pa | 91 | 5f2 6d1 7s2 |
| Neodymium | Nd | 60 | 4f4 6s2 | Uranium | U | 92 | 5f3 6d1 7s2 |
| Promethium | Pm | 61 | 4f5 6s2 | Neptunium | Np | 93 | 5f4 6d1 7s2 |
| Samarium | Sm | 62 | 4f6 6s2 | Plutonium | Pu | 94 | 5f6 7s2 |
| Europium | Eu | 63 | 4f7 6s2 | Americium | Am | 95 | 5f7 7s2 |
| Gadolinium | Gd | 64 | 4f7 5d1 6s2 | Curium | Cm | 96 | 5f7 6d1 7s2 |
| Terbium | Tb | 65 | 4f9 6s2 | Berkelium | Bk | 97 | 5f9 7s2 |
| Dysprosium | Dy | 66 | 4f10 6s2 | Californium | Cf | 98 | 5f10 7s2 |
| Holmium | Ho | 67 | 4f11 6s2 | Einsteinium | Es | 99 | 5f11 7s2 |
| Erbium | Er | 68 | 4f12 6s2 | Fermium | Fm | 100 | 5f12 7s2 |
| Thulium | Tm | 69 | 4f13 6s2 | Mendelevium | Md | 101 | 5f13 7s2 |
| Ytterbium | Yt | 70 | 4f14 6s2 | Nobelium | No | 102 | 5f14 7s2 |
Note: The lanthanide lutetium (Z = 71) and the actinide Lawrencium (Z = 103) are not f-block elements. They are listed under d-block elements.
Properties
All f-block elements form color complexes and salts but are lighter than those formed by d-block elements. The specific properties of lanthanides and actinides are below [1-4].
Lanthanides
- Soft metals whose hardness increases with increasing atomic number
- Silvery-white colored metals that tarnish when exposed to air and form oxides
- Melting points range from 1000 to 2000 K
- Good conductors of heat and electricity
- Non-radioactive in nature, except for promethium
- Possess a stable oxidation state of +3, so their fourth ionization energies tend to be extremely high.
- Good reducing agents
- Strongly paramagnetic, except La3+, Lu3+, Yb2+, and Ce4+, which are diamagnetic
- Reacts quickly with most nonmetals
- The atomic radius decreases from lanthanum to lutetium. It occurs due to a significant increase in nuclear charge and minimal additional electron shielding, allowing the nucleus to exert a greater force on the outer electrons. This phenomenon is known as lanthanide contraction.
- Used as a catalyst in the manufacture of petroleum and synthetic products
Actinides
- Properties are much more similar to the main group elements than the lanthanides.
- Soft, silvery-colored metals that can be cut easily with a knife
- Tarnish rapidly in the air
- Malleable and ductile
- High-density heavy elements with atomic mass ranging from 227 g/mol to 262 g/mol
- Highly reactive
- Radioactive in nature due to their large size. They do not have stable isotopes.
- Elements beyond uranium are synthetic and are artificially produced in the lab.
- Have complicated chemistry due to a wide range of oxidation states
- Atomic radius decreases from actinium to Lawrencium. This phenomenon is known as actinide contraction.
- Uranium and plutonium are used in the generation of nuclear energy. Americium is used in smoke detectors. Thorium is used in gas mantles. Actinium is used as a gamma source and neutron source. A large number of actinides are used in manufacturing atomic weapons.