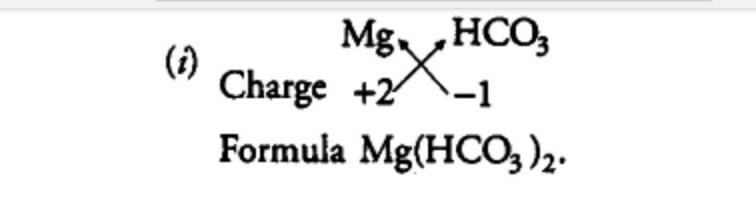Magnesium Bicarbonate
Magnesium bicarbonate, also known by its IUPAC name magnesium hydrogen carbonate, is a salt containing bicarbonate anion and magnesium cation represented by the formula C2H2MgO6 or Mg(HCO3)2 [1]. Since magnesium bicarbonate is unstable in a solid state, it exists in a dilute aqueous solution [2].
How is Magnesium Bicarbonate Prepared
It can be synthesized by treating magnesium acetate with sodium bicarbonate, involving the following reaction:
Mg(CH3COO)2 + 2NaHCO3 → Mg(HCO3)2 + 2CH3COONa
A solution of Mg(HCO3)2 commonly called magnesium bicarbonate water can be produced through the reaction of magnesium hydroxide (such as milk of magnesia) and pressurized carbon dioxide (like seltzer water) [4]:
Mg(OH)2 + 2CO2 → Mg(HCO3)2
The resulting solution when dried decomposes magnesium bicarbonate into magnesium carbonate, water, and carbon dioxide:
Mg + 2HCO3 → MgCO3 + CO2 + H2O
Properties and Characteristics of Magnesium Bicarbonate
General Properties |
|
| Molar Mass/Molecular Weight | 146.337 g/mol [1] |
Physical Properties |
|
| Color and Appearance | Light, white, unstable friable mass or powder [1, 2] |
| Odor | Odorless [2] |
| Melting Point | N/A |
| Boiling Point | 333.6 °C, 632.48 °F [2] |
| Charge | 0 (Mg2+ and HCO3–) [1] |
| State of matter at room temperature | Unstable in solid form but exists in the liquid state [2] |
| Solubility | Insoluble in ethanol [1] |
| Solubility in Water | Almost insoluble in water (0.077 g per 100 mL) [1] |
Uses
- Magnesium bicarbonate has numerous health benefits and is used for resisting changes in blood pH and keeping it within an optimal range [5].
- As an essential mineral, it helps in blood pressure regulation, protein synthesis, nerve and muscle functions, and blood glucose control in the human body [5].
- It is used as food additives like acidity regulators, alkali, anti-cracking and bleaching agents [1, 6].
- References
- Carbonic Acid, Magnesium Salt (2:1) – Pubchem.ncbi.nlm.nih.gov
- Magnesium Bicarbonate – Mg(HCO3)2 – Byjus.com
- Magnesium Bicarbonate – Chemspider.com
- Preparation of Solutions of Magnesium Bicarbonate for Deacidification – Jstor.org
- Magnesium Bicarbonate Water – The importance of bicarbonates and magnesium – Dancingwithwater.com
- Acidity Regulators – Chm.bris.ac.uk