Magnesium Nitrate
Magnesium nitrate represented by the chemical formula Mg(NO3)2 or MgN2O6 that bears the IUPAC name magnesium dinitrate [1] is a white crystalline hygroscopic powder that is soluble in water and alcohol [6]. It is a magnesium salt and contains chemical bonds that are ionic in nature [1].
Magnesium Nitrate Identification |
|
| CAS Number | 10377-60-3 [1] |
| PubChem CID | 25212 [1] |
| ChemSpider ID | 23415 [2] |
| EC Number | 233-826-7 [1] |
Composition and Synthesis
Magnesium nitrate can be prepared by combining magnesium sulfate with calcium nitrate [4].
MgSO4 + Ca(NO3)2 = Mg(NO3)2 + CaSO4
Properties and Characteristics of Magnesium Nitrate
General Properties |
||
| Molar mass/molecular weight | 148.313 g/mol [1] | |
Physical Properties |
||
| Color/appearance | White crystals [1] | |
| Melting point/freezing point | 89°C, 192.2°F [3] | |
| Boiling point | 330°C, 626°F [3] | |
| Density | 1.46 g cm-3 [3] | |
| State of matter at room temperature (normal phase) | Solid [1] | |
Chemical Properties |
||
| Solubility in water | 42 g/100 ml (at 20oC) [4] | |
| pH | 5-7 (acidic) [5] | |
Atomic Properties |
||
| Crystal structure | Cubic [6] | |
Prominent Reactions of Mg(NO3)2
A chemical reaction between magnesium nitrate and sodium carbonate produces magnesium carbonate and sodium nitrate [7].
Mg(NO3)2 + Na2CO3 = MgCO3 + 2NaNO3
Magnesium nitrate decomposes in water forming magnesium hydroxide, ammonia and oxygen [8].
Mg(NO3)2 + 4H2O = Mg(OH)2 + 2NH3 + 4O2
On heating Mg(NO3)2, it decomposes in magnesium oxide, nitrogen dioxide and oxygen [9].
2Mg(NO3)2 = 2MgO + 4NO2 + O2
It reacts with sodium hydroxide to give magnesium hydroxide and sodium nitrate [10].
Mg(NO3)2 + 2NaOH = Mg(OH)2 + 2NaNO3
Magnesium Nitrate Uses
- As a safe source of nitrate ions [4].
- In the production of nitrogenous fertilizers [4].
- In pyrotechnics and concentrated nitric acid manufacture [6].
- Helps in prilling and coating for ammonium nitrate production and is an ingredient of cosmetics and shampoos [6].
Is It Dangerous
Being a strong oxidizer, it may cause a fire hazard. It may also cause an explosion hazard when brought in contact with organic substances. Contact with eyes, skin and inhalation should be avoided as it results in irritation, coughing and shortness of breath. Ingestion in large quantities results in abdominal pain, dizziness, bloody diarrhea, vomiting, convulsions, weakness and collapse [6].
- References
- Magnesium Nitrate – Pubchem.ncbi.nlm.nih.gov
- Magnesium Nitrate – Chemspider.com
- Magnesium Nitrate – Americanelements.com
- Magnesium Nitrate – Sciencemadness.org
- Magnesium Nitrate Hexahydrate – Sigmaaldrich.com
- Magnesium Nitrate – Chemicalbook.com
- Question: Magnesium Nitrate and sodium carbonate Mg(NO_3)_2 + Na_2 – Chegg.com
- How is the balanced equation of magnesium nitrate plus water determined? – Quora.com
- The thermal decomposition of nitrate magnesium – Chemiday.com
- Question: Sodium hydroxide reacts with magnesium nitrate to form magnesium hydroxide – Chegg.com

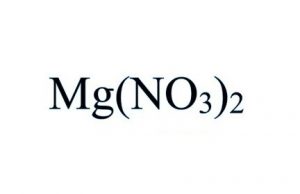
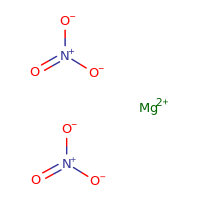





Magnesium nitrate is a hygroscopic magnesium salt, with the formula Mg(NO3)2, widely used as fertilizer.