Nucleophilic Acyl Substitution
What is Nucleophilic Acyl Substitution?
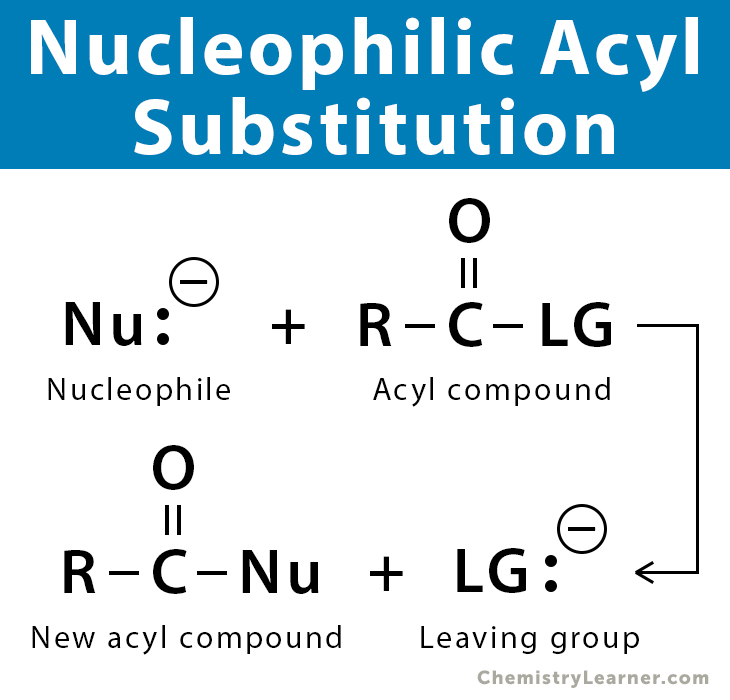
Nucleophiles are electron-rich species that are capable of donating electrons to an electrophile. They have a lone pair of electrons that attack electron-deficient sites in a molecule, thereby forming a new bond. Nucleophilic acyl substitution is a substitution reaction in which a nucleophile displaces a leaving group (LG) bonded to the central carbon of an acyl compound (RCO-LG) [1-8].
List of Acyl Compounds
The acyl compounds are carboxylic acid (RCO-OH) derivatives such as the following:
- Acyl halide (RCO-X)
- Acid anhydride (R(CO)O(CO)R’)
- Thioester (RCO-OSR’)
- Ester (RCO-OR’)
- Amide (RCO-NR’2)
Reactivity Order of the Acyl Compounds
Here is the order of reactivity of the acyl compound.
Acyl halide > acid anhydride > thioester > ester = carboxylic acid > amide > carboxylate
List of Nucleophiles
Here are some nucleophiles that add to the carbonyl (C=O) group.
Oxygen-centered: H2O, ROH, and OH–
Nitrogen-centered: R2NH, RNH-NH2, and HO-NH2
Carbon-centered: R3C-MgX, (R3C)2Cu-Li, and R3C-Li
Others: LiAlH4, NaBH4, X–, and HSO3–
Example of Nucleophilic Acyl Substitution
Fischer esterification of benzoic acid (C6H5COOH) with methanol (CH3OH) in the presence of sulfuric acid (H2SO4) gives methyl benzoate (C6H5COOCH3) is an example of nucleophilic acyl substitution [8-10].
C6H5COOH + CH3OH → C6H5COOCH3 + H2O
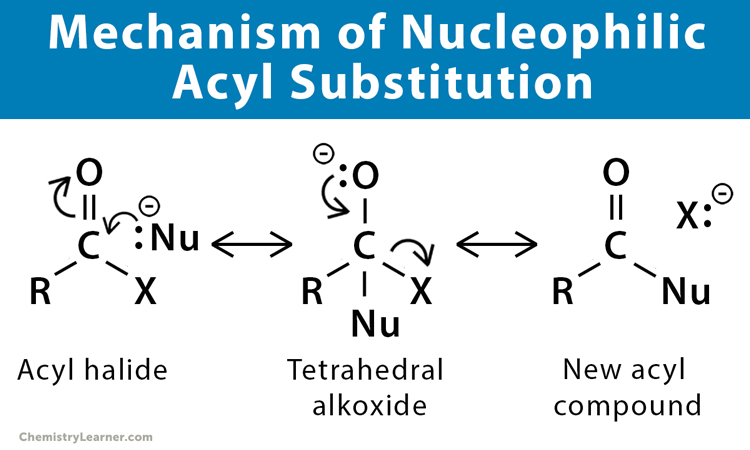
Mechanisms of Nucleophilic Acyl Substitution [1-7]
The mechanism of nucleophilic acyl substitution begins with a nucleophilic attack on an electrophilic carbonyl carbon, forming an intermediate known as tetrahedral alkoxide. Next, the negative charge on the alkoxide transfers to the leaving group. The second step involves eliminating the leaving group, allowing the C=O carbonyl bond to reform and create a new acyl compound.