Germanium
What is Germanium
Germanium (pronunciation: jer-MAY-ni-em) is a shiny, silvery element classified as a metalloid and represented by the chemical symbol Ge [1, 2]. As a relatively inactive element, germanium does not react with oxygen at 20°C and is insoluble in water but its compound, germanium dioxide, is slightly soluble in water [3].
Isotopes
Its five stable isotopes, germanium-70, germanium-72, germanium-73, germanium-74, and germanium-76, occur naturally out of which germanium-74 is the most common with a natural abundance of around 36% [4]. Germanium-76 is slightly radioactive with a half-life period of 1.6 X 1021 years and a natural abundance of around 7% [1, 4]. It also has more than 27 artificially produced radioisotopes whose atomic mass ranges from 58-89 [4].
Where is Germanium Found
Germanium rarely occurs as pure ore compounds and is found in small amounts in minerals like argyrodite and germanite [1]. While it can also be obtained from zinc ores, flue dust from zinc smelting is a commercial source of germanium [1]. Moreover, it can be recovered from coal combustion by-products [1].
The top 3 germanium producing countries include China, Russia, and Germany [1].
History
Origin of its Name: It is named after ‘Germania’, the Latin word for Germany [1].
Who discovered it: The German chemist Clemens A. Winkler is known to be the discoverer of germanium [1].
When and How was it Discovered
In September 1885, an unusual mineral ore was found by a miner who was working in the Himmelsfürst mine in the Freiberg district of Germany [1]. He passed it to Albin Weisbach, the German mineralogist who confirmed it was a new mineral, which we now know as Argyrodite (Ag8GeS6) [1]. His colleague Clemens Winkler analyzed the new mineral and found that it consisted of 18% sulfur, 75% silver, while the remaining 7% could not be explained [1, 5]. By February 1886, Winkler realized that it was a new element and named it germanium, the properties of which were earlier predicted by the Russian chemist Dmitri Mendeleev [1, 5].
Germanium Identification |
|||
| Atomic number | 32 [1] | ||
| CAS number | 7440-56-4 [1] | ||
| Position in the periodic table | Group | Period | Block |
| 14 [1] | 4 [1] | p [1] | |
Properties and Characteristics of Germanium
General Properties |
||||||||||||||||
| Relative atomic mass | 72.630 [1] | |||||||||||||||
| Atomic mass | 72.630 amu [1] | |||||||||||||||
| Molar mass | 72.6400 g/mol [6] | |||||||||||||||
| Allotropes | α-Ge, β-Ge [1] | |||||||||||||||
Physical Properties |
||||||||||||||||
| Color | Silvery-white [1, 6] | |||||||||||||||
| Melting point/freezing point | 938.25 °C, 1720.85 °F [1] | |||||||||||||||
| Boiling point | 2833 °C, 5131 °F [1] | |||||||||||||||
| Density | 5.3234 g cm-3 [1] | |||||||||||||||
| State of matter at room temperature (solid/liquid/gas) | Solid [1, 5] | |||||||||||||||
| Hardness | ||||||||||||||||
| – Brinell | 7273.4 MPa [7] | |||||||||||||||
| – Mohs | 6 [7] | |||||||||||||||
| – Vickers | 8012.03 MPa [7] | |||||||||||||||
| Electrical conductivity | 2000 S/m [7] | |||||||||||||||
| Charge | +4, +2 [8] | |||||||||||||||
| Thermal (heat) conductivity | 60 W/(m K) [7] | |||||||||||||||
| Specific heat | 320 J kg-1 K-1 [1] | |||||||||||||||
| Bulk modulus | Unknown [1] | |||||||||||||||
| Shear modulus | Unknown [1] | |||||||||||||||
| Young’s modulus | Unknown [1] | |||||||||||||||
| Vapor pressure | ||||||||||||||||
| – Temperature (K) | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 | 2200 | 2400 | |||||
| – Pressure (Pa) | – | – | – | – | – | – | – | – | – | – | – | |||||
Chemical Properties |
||||||||||||||||
| Oxidation state/Oxidation number | −4, (−3), (−2), (−1), +1, +2, +3, +4 [1] | |||||||||||||||
| Isotopes | Isotope | Mass | Abundance (%) | Half-life | Mode of decay | |||||||||||
| 70Ge | 69.924 | 20.57 | – | – | ||||||||||||
| 72Ge | 71.922 | 27.45 | – | – | ||||||||||||
| 73Ge | 72.923 | 7.75 | > 1.8 X 1023 y | β- | ||||||||||||
| 74Ge | 73.921 | 36.5 | – | – | ||||||||||||
| 76Ge | 75.921 | 7.73 | 1.6 X 1021 y | β-β- | ||||||||||||
Atomic Data of Germanium (Element 32)
| Valence electrons | 4 [9] | |||||||
| Quantum numbers | ||||||||
| – n | 4 [10] | |||||||
| – ℓ | 1 [10] | |||||||
| – mℓ | 0 [10] | |||||||
| – ms | +1/2 [10] | |||||||
| Electron configuration (noble gas configuration) | [Ar] 3d104s24p2 [1] | |||||||
| Atomic structure | ||||||||
| – Number of electrons | 32 [5] | |||||||
| – Number of neutrons | 42 [5] | |||||||
| – Number of protons | 32 [5] | |||||||
| Radius of Atom | ||||||||
| – Atomic radius | 2.11 Å [1] | |||||||
| – Covalent radius | 1.20 Å [1] | |||||||
| Electronegativity (Pauling-scale) | 2.01 [1] | |||||||
| Electron affinity | 118.939 kJ mol-1 [1] | |||||||
| Ionization energy (kJ mol-1) | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
| 762.179 | 1537.456 | 3302.124 | 4410.644 | 9021.4 | – | – | – | |
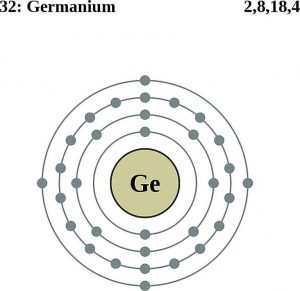
Germanium Electron Configuration (Bohr Model)
What is it Used for
- Pure germanium was usually doped with gallium, arsenic, or other elements for use as a semiconductor in transistors and integrated circuits of electronic applications [1, 5]. Today, it has been replaced by other semiconductors [1].
- Because of its high dispersion and refraction index, germanium is commonly used in objective lenses for microscopes and wide-angle lenses for camera [1].
- It is added to alloys, including silver, for stopping it from tarnishing [1]. It can as well be used as a catalyst and in fluorescent lamps [1, 5].
- Since germanium and its oxide are transparent in infrared wavelength, they make excellent infrared optical material and are utilized in infrared spectroscopes [1].
- Organic germanium, sold as supplements, is believed to be useful for promoting a healthy immune system, destroying free radicals, and supplying oxygen in the body [11]. It is claimed to be a remedy for health conditions like allergies, arthritis, HIV, cancer, and asthma [11].
- It is used in making solar cells that are placed in solar panels [6].
Is Germanium Toxic
The element 32 is considered non-toxic [1]. Although some of its compounds are slightly toxic in mammals, they are known for their anti-bacterial properties for which researchers are now looking into their probable use in medications [1].
Interesting Facts
- Germanium’s graphical representation features an image of a transistor, which indicates the early use of the element [1].
- Being a metalloid, germanium has the properties of both nonmetals and metals [6].
- Like water, germanium is known to expand on freezing [6].
- It was commonly used in high-resolution radar during the Second World War [6].
Germanium Cost
The cost of pure germanium is around $3.60 per gram, and in bulk, its price is around $1.20 per gram [5].
- References
- http://www.rsc.org/periodic-table/element/32/germanium
- https://education.jlab.org/itselemental/ele032.html
- https://pubchem.ncbi.nlm.nih.gov/compound/Germania#section=Top
- https://education.jlab.org/itselemental/iso032.html
- https://www.chemicool.com/elements/germanium.html
- https://www.livescience.com/29520-germanium.html
- http://periodictable.com/Elements/032/data.html
- Lenntech.com
- http://hyperphysics.phy-astr.gsu.edu/hbase/Solids/sili.html
- http://chemistry-reference.com/q_elements.asp?Symbol=Ge&language=en
- https://www.healthline.com/health/is-germanium-a-miracle-cure