Adamantane
Adamantane is a chemical compound that is quite useful in some technological as well as medicinal applications. Read this article to know more about this chemical.
What is Adamantane
It is a crystalline chemical compound. It is a cycloalkane (alkanes having rings of carbon atoms in their molecular structure) and is the simplest diamondoid (structures having resemblance to diamond). It is for this reason that this compound has been named “Adamantane”.
Adamantane Chemical Formula
This chemical compound has the formula of C10H16.
Adamantane History and Synthesis
H. Decker was the first person who, at a 1924 conference, suggested that a Hydrocarbon with the chemical formula of C10H16 and a molecular structure like a diamond may exist.] He named this molecule “Decaterpene”.
In the same year, the German chemist Hans Meerwein tried to synthesize the compound by reacting Formaldehyde and Diethyl Malonate under the influence of Piperidine. It was the first laboratory-attempt to synthesis this substance. But instead of the desired product Meerwein got 1, 3, 5, 7-tetracarbomethoxybicyclo [3.3.1] nonane-2, 6-dione. The newfound compound was named “Meerwein’s Ester”. It was later used in the synthesis process of Adamantane and all its derivatives. Another German chemist named D. Bottger attempted to obtain the same compound using Meerwein’s Ester but got another derivative of it.
Vladimir Prelog was the first person to successfully synthesize Adamantane from Meerwein’s Ester in 1941. But the process followed by Prelog was too complicated for regular use. In 1956, the process was refined and simplified but it was still not simple enough for regular use. In 1959, Paul von Ragué Schleyer accidentally discovered a much simpler process to produce this substance.
In this process, Dicyclopentadiene was hydrogenated under the influence of a catalyst (e.g. Platinum Dioxide). Then, the resultant product was transformed into Adamantane in the presence of another catalyst (some Lewis acid like Aluminum Chloride). The substance produced in this process was very affordable.
All the mentioned processes produce a polycrystalline powder (solid composed of many small, almost always microscopic crystals) of the compound. It is possible to create single crystals from this powder.
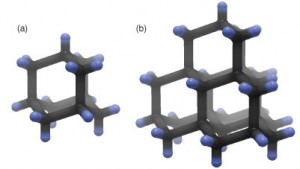
Picture 1 – Adamantane
Source – llnl.gov
Adamantane Natural Occurrence
Before Adamantane was obtained through synthesis process, Czech chemists S. Landa and V. Machacek isolated this substance from petroleum in the year 1933. It was done with the help of fractional distillation. Petroleum was heated to a point where only solid impurities were left in it. The ensuing steam was sent through a Fractional Distillation Column. The Column temperature decreased because of the steam. As a result, Hydrocarbon fractions condensed all along the Column according to their different boiling points. Only a few milligram of the substance was produced in this process. But the high melting and boiling points of the compound were noticed.
Adamantane Structure
A molecule of this compound contains three Cyclohexane rings (a Cycloalkane with chemical formula of C6H12). These are organized in the “armchair” or “chair” conformation (form of stereoisomerism).
The length of the Carbon-Carbon bond in the molecular structure is 1.54 Å (Angstrom.1 Å = 1/10,000,000,000 meter). It is almost identical to the carbon- carbon bond in a diamond molecule. The distance between the Carbon and the Hydrogen in the molecule is 1.112 Å.
Adamantane Nomenclature
According to the systematic nomenclature rule, the right name for this substance should be tricyclo[3.3.1.13,7]decane. But International Union of Pure and Applied Chemistry (IUPAC) recommend continuing using “Adamantane” as its name.
Adamantane Molecular Weight
The molecular weight of this substance is 136.234 g/mol.
Adamantane Properties
Following are the basic and physical properties of this compound:
- It has the physical state of a colorless solid.
- The melting point of this compound is 270 °C. It is higher than all the other Hydrocarbons with similar molecular weight.
- The density of this substance is 1.07 g/cm3.
- The odor of this chemical compound resembles that of the camphor (a white solid with an aromatic smell).
- This substance is insoluble in water. But it has high solubility in a number of common organic liquids.
- Water Vapor Distillation process (distillation with the help of water or steam) can be applied to this compound.
- It has good thermal stability.
Adamantane Derivatives
Following are the names of some of the derivatives of this chemical compound:
- 1-Adamantyl methyl ketone (1-Acetyladamantane) or C12H18O
- 2-Adamantanone or C10H14O
- 2-Adamantanol (2-Hydroxyadamantane) or C10H16O
- 1- Amino -3-Hydroxy –Adamantane or C10H17NO
- 2-Aminoadamantane Hydrochloride or C10H17NHC
- 1-Acetamidoadamantane or C12H19NO
- 1-Aminoadamantane Sulfate ( Bis[1-Aminotricyclo (3.3.1.1.3.7)decane]sulfate) or (C10H17N)2H2SO4
- 1-Chloro-3,5- Dimethyladamantane or C12H19Cl
- 1-Adamantanemethanol (1-Adamantylmethanol) C11H18O
- 1,3-Dihydroxyadamantane (1,3-Adamantanediol) C10H16O2
Adamantane as an Antiviral Medicine
A pure form of this substance is not used in drugs or for other medicinal purposes. The different derivatives of this compound are usually used for the treatment of various diseases. Amantadine was the first derivative to be used as an antiviral drug for the treatment of flu and later for Parkinson’s disease. Some other derivative drugs are:
- Memantine
- Rimantadine
- Dopamantin
- Tromantadine
- Vildagliptin
- Karmantadin
Adamantane polymers have been patented to be used as antiviral drug against HIV. H1N1 or Swine Flu viruses are capable of resisting these antiviral compounds.
Other Uses of Adamantane
Being an unfunctionalized hydrocarbon, the compound is not very useful. The few uses of this substance are mentioned below:
- It is used in polymer formulations and dry etching masks.
- Adamantane is considered to be a common standard for Chemical Shift referencing in Solid-State NMR spectroscopy.
- It is sometimes used to elongate the life of Gain Medium in Dye Lasers.
Adamantane MSDS
This compound can be harmful for humans in case of over exposure. It may cause the following problems for humans:
Eye Irritation
It may cause eye irritation in case of direct contact. In this case, the eyes should be washed with a lot of water. The victim should immediately seek medical consultancy.
Skin Irritation
It may also cause skin irritation. The affected area should be washed immediately with soap and plenty of water. The infected clothes should be removed and washed properly before re-use.
Health problems on Ingestion
If the compound is ingested by accident, the victim should immediately take medical assistance. It is advisable to rinse the mouth and drink 2-4 glasses of water or milk to dilute the material. Vomiting should not be induced.
Respiratory difficulties on Inhalation
In case of breathing difficulty, Oxygen must be provided. If the victim is not breathing, artificial respiratory must be applied. Getting immediate medical aid is important.
- References
- http://www.drugs.com/drug-class/adamantane-antivirals.html
- http://www.sciencedirect.com/science/article/pii/0378381295029494
- http://www.chemeurope.com/en/encyclopedia/Adamantane.html




