Actinides
Definition: What are actinides?
Actinides belong to the family of inner transition metals with partially filled 5f subshell. They are named after actinium, the first element in the series, and play a crucial role in nuclear chemistry [1].
Where are the actinides located in the periodic table?
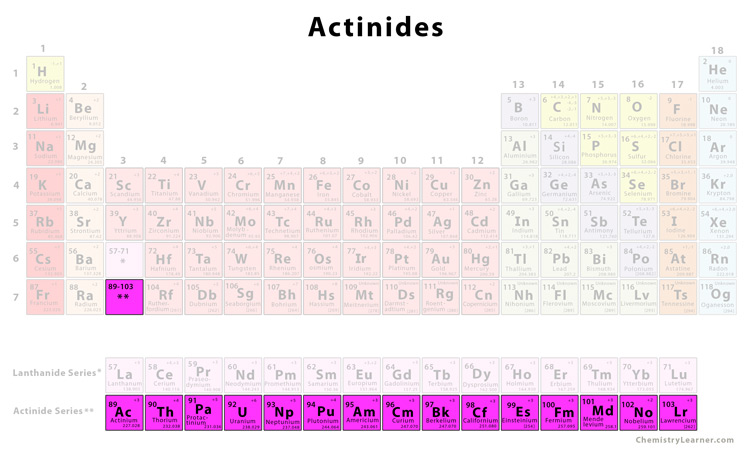
The actinides are located in group 3 of the 7th period of the periodic table in between radium (Ra) and Rutherfordium (Rf). They are shown below the body of the periodic table for simplicity. They are also known as f-block elements since the 5f shell is getting filled progressively as the atomic number changes from 89 to 103. The actinides have a general electronic configuration given by [Rn] 5f1-146d0-17s2, where [Rn] is the electronic configuration of the nearest noble gas radon [2].
List of actinides [3]There are a total of 15 actinides in the periodic table. |
||
| Element | Symbol | Atomic Number |
| Actinium | Ac | 89 |
| Thorium | Th | 90 |
| Protactinium | Pa | 91 |
| Uranium | U | 92 |
| Neptunium | Np | 93 |
| Plutonium | Pu | 94 |
| Americium | Am | 95 |
| Curium | Cm | 96 |
| Berkelium | Bk | 97 |
| Californium | Cf | 98 |
| Einsteinium | Em | 99 |
| Fermium | Fm | 100 |
| Mendelevium | Md | 101 |
| Nobelium | No | 102 |
| Lawrencium | Lr | 103 |
Common Properties and Characteristics of the Actinides
Actinides have some properties that are similar to one another, with some having properties similar to d-block elements
Physical Properties of Actinides [4, 5]
- Silvery or silvery-white metals in their luster form
- Soft metals which can be easily cut with a knife
- Are malleable and ductile
- Very dense metals with distinctive structures, i.e., have several allotropes (e.g., Pu has 6)
- Have higher melting and boiling points than lanthanides
Chemical Properties of Lanthanides [4, 5]
- Many are synthetically prepared in the laboratory by using particle accelerators
- Radioactive elements due to instability of the isotopes
- Highly electropositive elements with high reactivity
- Tarnish in air and display pyrophoric characteristics, i.e., spontaneously ignite in air
- React with boiling water and acid to release hydrogen gas
- Form stable complexes with nonmetals and ligands such as chloride, sulfate, carbonate, and acetate
- Display several oxidation states, with +4 being most common during the formation of oxides
- Are highly toxic
- Are paramagnetic
Uses and Applications of Actinides [6]
- As fuel in nuclear reactors (e.g., plutonium, uranium, and thorium)
- In nuclear weapons (e.g., plutonium and uranium)
- In smoke detectors (e.g., Americium) and gas mantles (e.g., thorium)
- As a neutron source and as a gamma source in industry and medicine (e.g., actinium)
Actinide Contraction
Actinide contraction is a phenomenon in which the atomic radii of the actinides decrease as the atomic numbers increase across the series. This phenomenon is because as more and more electrons are added to the atom, the 5f electrons are incapable of providing perfect shielding from the nucleus to the 7s electrons. As a result, the effective nuclear charge increases, and the 6s electrons come closer to the nucleus, thereby decreasing the atomic radius [7].
FAQs
Q.1. Why are actinides radioactive?
Ans. Actinides are radioactive because of their unstable nuclei.
Q.2. Are actinides rare-earth?
Ans. Actinides are not considered to be rare earth as most of them are not available in nature and are artificially synthesized in the laboratory.
Q.3. Which actinides occur naturally on Earth?
Ans. The two actinides that occur naturally in the earth’s crust are thorium and uranium. Besides, actinium and protactinium are also available in a tiny amount.
Q.4. How are actinides extracted?
Ans. Uranium and thorium can be easily extracted from the earth’s crust from their fluorides, which are reduced with calcium by redox reactions.
References
- Definition of actinides – Britannica.com
- Location of actinides – Byjus.com
- List of actinides – Thoughtco.com
- Properties of actinides – Chem.libretexts.org
- Properties of actinides – Thoughtco.com
- Applications of actinides – Vedantu.com
- Actinide contraction – Shaivikchemistryclasses.com