Antimony
What is Antimony
Antimony (pronunciation: AN-te-MOH-nee) is a lustrous metallic element that belongs to the family of metalloids and is represented by the chemical symbol Sb [1, 2, 3]. There are two stable isotopes of antimony, including 121Sb and 123Sb with a natural abundance of 57.21% and 42.79% respectively [4]. It also has 35 artificially-produced radioactive isotopes, out of which the longest-lived are 125Sb, 124Sb, and 126Sb with half-lives of 2.75856 years, 60.2 days, and 12.35 days respectively [4].
Where is Antimony Found
Antimony occurs naturally in trace amounts in more than 100 types of mineral ores, mainly from antimony (III) sulfide (stibnite) [1, 5]. It is commercially obtained by roasting antimony trisulfide to form antimony oxide and then reducing it with carbon [1]. Antimony is also found in its native form [1]. China, Bolivia, and Tajikistan are the top 3 producers while China, Russia, and Bolivia have the largest antimony reserves in the world [1].
History
Origin of its Name: The name is derived from ‘anti-monos’, a Greek word that means “opposed to solitude” or “against solitude” [1, 6].
Who discovered it: Since a 5000-year-old vase made of pure antimony is shown in the Louvre museum, the element was probably found by the ancient people thousands of years ago [1].
When and How was it Discovered
The early use of antimony sulfide (Sb2S3) has been recorded in an Egyptian papyrus dating back to 1600 BC [1]. Its mineral ore, stibnite, was commonly used in its black form as mascara by the ancient people notably by the temptress Jezebel [1].
Lead antimonate, a yellowish pigment was used by the Chaldean craftsmen during 600-500 BC [1]. It was used in the thin coating of ornamental bricks found at Babylon made during the reign of Nebuchadnezzar from 604 to 561 BC [1].
In the Medieval times, it was widely used for hardening lead while some were taken as a laxative [1].
Antimony Identification |
|||
| Atomic number | 51 [1] | ||
| CAS number | 7440-36-0 [1] | ||
| Position in the periodic table | Group | Period | Block |
| 15 [1] | 5 [1] | p [1] | |
Properties and Characteristics of Antimony
General Properties |
||||||||||||||||
| Relative atomic mass | 121.760 [1] | |||||||||||||||
| Atomic mass | 121.760 amu [1] | |||||||||||||||
| Molar mass | 121.7600 g/mol [7] | |||||||||||||||
| Molecular weight | 121.76 g/mol [2] | |||||||||||||||
| Allotropes | White Sb, Black Sb, Yellow Sb [1] | |||||||||||||||
Physical Properties |
||||||||||||||||
| Color | Silvery-white [2, 8] | |||||||||||||||
| Melting point/freezing point | 630.628 °C, 1167.13 °F [1] | |||||||||||||||
| Boiling point | 1587 °C, 2889 °F [1] | |||||||||||||||
| Density | 6.68 g cm-3 [1] | |||||||||||||||
| State of matter at room temperature (solid/liquid/gas) | Solid [1, 8] | |||||||||||||||
| Hardness | ||||||||||||||||
| – Brinell | 294 MPa [9] | |||||||||||||||
| – Mohs | 3 [9] | |||||||||||||||
| – Vickers | Unknown [9] | |||||||||||||||
| Electrical Conductivity | 2.5 X 106 S/m [9] | |||||||||||||||
| Charge | -3, +5 [10] | |||||||||||||||
| Thermal (heat) conductivity | 24 W/(m K) [9] | |||||||||||||||
| Specific heat | 207 J kg-1 K-1 [1] | |||||||||||||||
| Bulk modulus | 42 GPa [1] | |||||||||||||||
| Shear modulus | Unknown [1] | |||||||||||||||
| Young’s modulus | Unknown [1] | |||||||||||||||
| Vapor pressure | ||||||||||||||||
| – Temperature (K) | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 | 2200 | 2400 | |||||
| – Pressure (Pa) | – | – | – | – | – | – | – | – | – | – | – | |||||
Chemical Properties |
||||||||||||||||
| Oxidation state/Oxidation number | −3, +3, +5 [1] | |||||||||||||||
| Isotopes | Isotope | Mass | Abundance (%) | Half-life | Mode of decay | |||||||||||
| 121Sb | 120.904 | 57.21 | – | – | ||||||||||||
| 123Sb | 122.904 | 42.79 | – | – | ||||||||||||
Atomic Data of Antimony (Element 51)
| Valence electrons | 5 [11] | |||||||
| Quantum numbers | ||||||||
| – n | 5 [11] | |||||||
| – ℓ | 1 [11] | |||||||
| – mℓ | 1 [11] | |||||||
| – ms | +1/2 [11] | |||||||
| Electron configuration (noble gas configuration) | [Kr] 4d105s25p3 [1] | |||||||
| Atomic structure | ||||||||
| – Number of electrons | 51 [8] | |||||||
| – Number of neutrons | 70 [8] | |||||||
| – Number of protons | 51 [8] | |||||||
| Radius of Atom | ||||||||
| – Atomic radius | 2.06 Å [1] | |||||||
| – Covalent radius | 1.40 Å [1] | |||||||
| Electronegativity (Pauling-scale) | 2.05 [1] | |||||||
| Electron affinity | 100.924 [1] | |||||||
| Ionization energy (kJ mol-1) | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
| 830.583 | 1604.55 | 2441.1 | 4264.7 | 5403 | 10420 | – | – | |
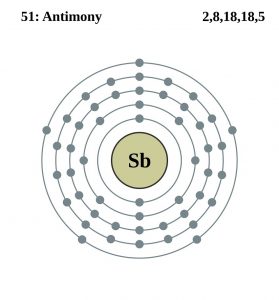
Antimony Electron Configuration (Bohr Model)
What is it Used for
- It is used for producing some semiconductor devices, including diodes and infrared detectors [1].
- It is mixed with lead and other metals to form alloys that increase the metals’ strength, hardness, and durability [1, 3]. While antimony-lead alloy is commonly used in car batteries, antimony alloys are also used in cable sheathing, casting bullets, and making type characters in printing presses [1].
- Compounds of antimony are used for manufacturing flame-resistant materials, paints, enamels, pottery and glass [1].
- An alloy of antimony, lead, tin, and copper, called Babbit metal, is used in making machinery bearings for its hardness and slipperiness [8].
Antimony Toxicity
It occurs either during the treatment of parasitic diseases, including schistosomiasis and leishmaniasis or because of occupational exposure [12]. Also, antimony trioxide is considered to be carcinogenic to humans [12]. Exposure to antimony compounds like antimony trisulfide, antimony tribromide, antimony pentasulfide, etc. may cause respiratory, gastrointestinal, cardiovascular, reproductive, and dermal effects [12].
Interesting Facts
- The stibnite crystals, resembling a group of swords and made from sulfur and antimony, were auctioned on 2nd June 2013 with an opening bidding price of $32,500 [5]. The mineral, measuring 9 X 10 X 4 inches, was obtained from the Lushi Mine, China [5].
- Antimony is graphically represented by the Eye of Horus, a symbol of royal power, good health, and protection from ancient Egypt [1]. It also indicates the use of antimony sulfide as eye makeup [1].
- It is stable in air and water, being slightly oxidized by air and insoluble in water [2].
- In the pure form, it is neither ductile nor malleable like true metals [14].
Price of Antimony
The cost of pure antimony is about $0.045 per gram, and in bulk, it costs about $0.0044 per gram [8]. In July 2018, the average price of antimony per metric ton is $8,294 [13].
- References
- http://www.rsc.org/periodic-table/element/51/antimony
- https://pubchem.ncbi.nlm.nih.gov/compound/antimony
- https://education.jlab.org/itselemental/ele051.html
- https://education.jlab.org/itselemental/iso051.html
- https://www.livescience.com/37390-antimony.html
- https://chemaust.raci.org.au/article/julyaugust-2019/antimony.html
- https://www.webqc.org/molecular-weight-of-Sb(antimony).html
- https://www.chemicool.com/elements/antimony.html
- http://periodictable.com/Elements/051/data.html
- https://www.learner.org/wp-content/interactive/periodic/groups7.html
- http://chemistry-reference.com/q_elements.asp?Symbol=Sb
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3037053/
- http://usantimony.com/pricing.htm
- http://scienceviews.com/geology/antimony.html










Thanks