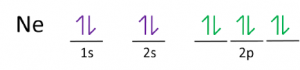Neon
What is Neon
Neon (NEE-on) is classified as a non-metal, represented by the chemical symbol Ne, belonging to the noble gas family. A highly inert and diamagnetic element, it does not react with any other element or compound. Out of its 14 isotopes, only Ne-20, Ne-21, and Ne-22 are naturally occurring with a stable half-life [1, 2].
Where is Neon Found in Nature
It occurs naturally in the Earth’s atmosphere at a concentration of 18 parts per million. On a commercial scale, it is obtained by fractional distillation of liquid air [1].
History
Origin of its Name: The name is derived from the Greek word ‘neos’ that means new [1].
Who Discovered Neon: Morris Travers and Sir William Ramsay [1]
When, Where, and How was it Discovered
In 1898, after obtaining krypton gas from liquid argon, both Ramsay and Travers repeated the experiment once more to find out a lighter gas that would be positioned above the former element. This time the solid argon was made to evaporate slowly under low pressure to isolate the gas coming out first. When they successfully managed to do so, the new gas was tested in their spectrometer that exhibited brilliant red glow. It was Ramsay who named it neon [1].
Identification |
|||
| Atomic number | 10 [1] | ||
| CAS number | 7440-01-9 [1] | ||
| Position in the periodic table [1] | Group | Period | Block |
| 18 | 2 | p | |
Classification, Properties and Characteristics of Neon
General Properties |
||
| Relative/Average atomic mass | 20.180 [1] | |
| Atomic mass/weight | 20.180 atomic mass units [4] | |
| Molar mass/Molecular weight | 20.18 g/mol [5] | |
| Mass number | 20 | |
Physical Properties |
||
| Color/physical appearance | Colorless [1] | |
| Odor | Odorless [6] | |
| Melting point/freezing point | -248.59°C (-415.46°F) [1] | |
| Boiling point | -246.046°C (-410.883°F) [1] | |
| Density | 0.000825 g/cm3 [1] | |
| Hardness | Unknown [7] | |
| Standard/Physical state of matter at normal room temperature (solid/liquid/gas) | Gas [1] | |
| Electrical conductivity | Unknown [3] | |
| Thermal conductivity | 0.05 Wm-1K-1 [3] | |
Chemical Properties |
||
| Flammability | Non-flammable [8] | |
| Oxidation state/Oxidation number | 0 [1] | |
Atomic Data of Neon (Element 10)
| Valence electrons | 8 [9] | ||||||
| Electron configuration (noble gas configuration) | [He] 2s22p6[1] | ||||||
| Atomic structure [3] | |||||||
| – Number of Electrons | 10 | ||||||
| – Number of Neutrons | 10 | ||||||
| – Number of Protons | 10 | ||||||
| Radius of atom | |||||||
| – Atomic radius | 1.54 Å [1] | ||||||
| – Covalent radius | 0.62 Å [1] | ||||||
| Electronegativity | Unknown [10] | ||||||
| Ion charge | Unknown [10] | ||||||
| Ionization energy [1]
(kJmol-1) |
1st | 2nd | 3rd | 4th | 5th | 6th | 7th |
| 2080.662 | 3952.325 | 6121.99 | 9370.66 | 12177.41 | 15237.93 | 19999.086 | |
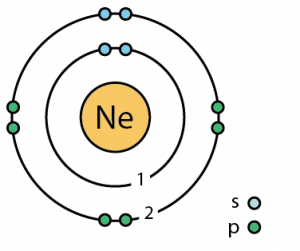
Neon Atomic Structure (Bohr Model)
How Commonly is Neon Used
- Red lights made by filling vacuum discharge tubes with the neon are used in display advertising signs [1].
- A combination of Ne and helium is used in the manufacture of highly stable, low-cost lasers that have several industrial and laboratory applications [2, 11].
- Neon lamps are used in high-voltage switching gear and indicators, and lightning arrestors to maintain the safety of electrical installation [1].
- Neox, a mixture of Ne and oxygen is sometimes used in the breathing apparatus of scuba and deep sea divers [12].
- Liquid neon is an effective cryogenic refrigerant with a higher refrigerating capacity than liquid hydrogen and helium [1].
Is the Element Toxic
No scientific studies have been able to categorize Ne as a poisonous gas. However, it could act as an asphyxiant causing dizziness, suffocation, dullness, and headache [13].
Interesting Facts
- Ionized Ne can pass through water fog without any obstruction [14].
- Although the inert gas does not react with other elements, it may form a compound with fluorine [15].
- References
- http://www.rsc.org/periodic-table/element/10/neon
- https://education.jlab.org/itselemental/ele010.html
- https://www.chemicool.com/elements/neon.html
- http://www.chemicalelements.com/elements/ne.html
- https://www.gordonengland.co.uk/elements/ne.htm
- http://www.elementalmatter.info/neon-properties.htm
- http://periodictable.com/Properties/A/MohsHardness.v.html
- https://cameochemicals.noaa.gov/chemical/7169
- https://courses.lumenlearning.com/cheminter/chapter/electron-dot-diagrams/
- http://schools.cbe.ab.ca/b858/dept/sci/teacher/zubot/Sci10ws/Chapter8/2Commonion.htm
- http://www.physics-and-radio-electronics.com/physics/laser/heliumneonlaser.html
- Envinsci.co.uk
- https://www.cdc.gov/niosh/hhe/reports/pdfs/2001-0081-2877.pdf
- https://www.thoughtco.com/interesting-neon-element-facts-4077247
- https://education.jlab.org/itselemental/ele010.html