Octet Rule
What is the Octet Rule
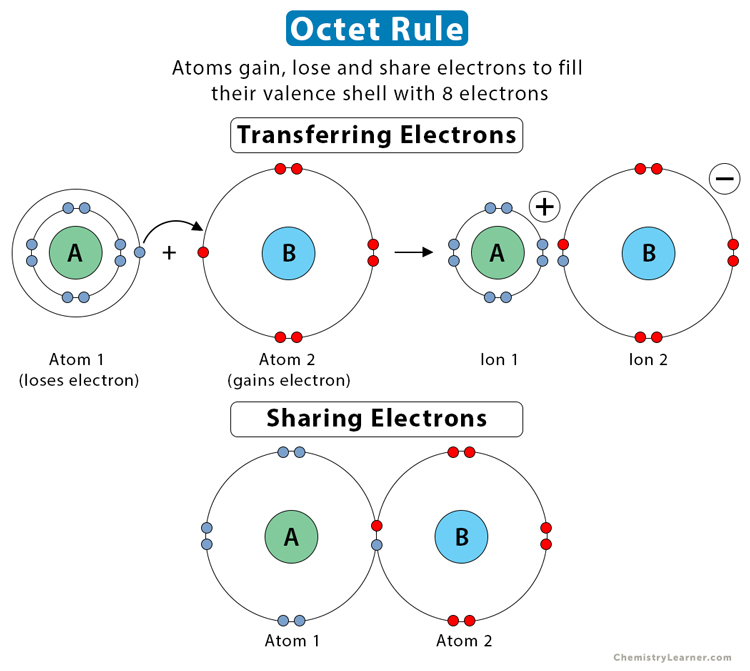
Atoms are not stable by themselves. The octet rule states that the number of electrons in an atom’s outermost or valence shell must be eight to attain stability. During chemical bonding, atoms gain, lose, or share electrons and become stable. In an ionic bond, atoms transfer electrons and become ions. Atoms share electrons in a covalent bond [1-4].
On the other hand, stable and inert noble gases have eight valence electrons, except helium, with a valence shell electron configuration given by ns2np6. Upon forming a chemical bond, atoms acquire the electron configurations of their nearest noble gas. The octet rule is important because it can explain a chemical bond based on an atom’s outermost electron configuration.
American chemist Gilbert Lewis studied the properties of ions and molecules. In 1916, he formulated the octet rule in his cubic atomic theory.
Which Elements Follow the Octet Rule
The octet rule is satisfied by s- and p-block elements of the periodic table collectively known as main group elements. The s-block elements include groups 1, 2, and helium, and the p-block elements include groups 13 to 18, except helium. Three main group elements, hydrogen, helium, and lithium, do not follow the octet rule. On the other hand, the d- and f-block elements, like transition and inner transition metals, also do not obey the octet rule [1-4].
Octet Rule Examples [5]
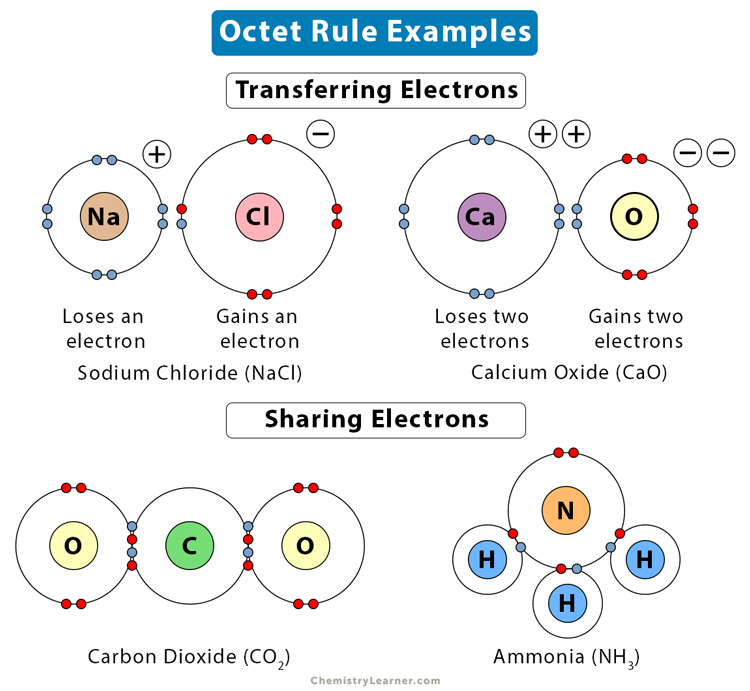
1. Sodium Chloride (NaCl)
The octet rule can explain the formation of sodium chloride (NaCl) through electron transfer, resulting in an ionic bond. Sodium (Na) has one electron in its valence shell. It will lose the electron and become sodium ion (Na+). The electron configuration of Na+ is the same as that of neon, which is [He]2s22p6. Chlorine (Cl) has seven electrons in its valence shell. It will accept the electron from Na, transform to chloride ion (Cl–), and take the electron configuration of argon, which is [Ne]3s23p6. The chemical bonding in NaCl is shown in the image below.
2. Carbon Dioxide (CO2)
The octet rule can also explain the covalent bonding of carbon dioxide (CO2) through sharing of electrons. Carbon (C) has four electrons in its valence shell, and oxygen (O) has six. The electronegativity difference between C and O predicts that the chemical bonding in CO2 is covalent. Hence, C and O will share two valence electrons and form two double bonds, as shown in the image below. Thus, in CO2, both C and O have eight outermost electrons.
3. Ammonia (NH3)
The octet rule can also explain the covalent bonding in ammonia (NH3). Nitrogen (N) has five electrons in its valence shell and requires three more to complete its octet. Hydrogen (H) will share its lone electron with nitrogen, and three hydrogen atoms are required to complete nitrogen’s outermost shell. Thus, N and H will form three covalent bonds, as shown in the image below.
The following image shows a few examples of molecules where the octet rule is followed.
FAQs
Ans. Calcium has two electrons in its valence shell. Therefore, it will donate those two electrons and form an ionic bond. For example, in calcium oxide (CaO), Ca loses two electrons to oxygen (O) becomes Ca2+. It takes the electron configuration of argon, which is [Ne]3s23p6.
Ans. Yes. Potassium (K) has one valence electron and will give it to iodine (I), which has seven. Thus, by forming an ionic bond, both K and I satisfy the octet rule.
Ans. The main difference between the octet and duplet rule is that the octet rule states that an atom or an ion can have a maximum of eight electrons in its valence shell. On the other hand, the duplet rule states that an atom can have a maximum of two electrons in its valence shell.