Zirconium
What is Zirconium
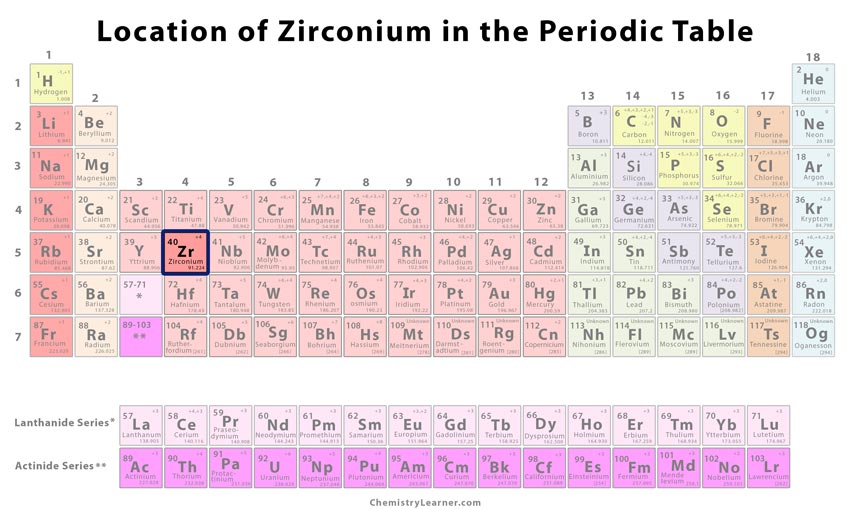
Zirconium (pronunciation: zer-KO-nee-em) is a hard, silvery-gray element that belongs to the group of transition metals and is represented by the chemical symbol Zr [1, 2, 3]. It is a malleable and ductile metallic element that can readily react with other elements to form stable compounds [2].
Isotopes of Zirconium
Zirconium has four stable, naturally occurring isotopes, including 90Zr, 91Zr, 92Zr, and 94Zr with a natural abundance of 51.45%, 11.22%, 17.15%, and 17.38% respectively [4]. There are roughly 29 radioisotopes out of which the two most stable are 96Zr and 93Zr with half-lives of about 2.0 X 1019 years and 1.53 million years respectively [4].
Where is Zirconium Found
Zirconium is naturally found in several mineral species, mainly in zircon (zirconium silicate or ZrSiO4) and baddeleyite (zirconium dioxide or zirconia or ZrO2) [1, 2]. The metal is commercially isolated from zircon by first converting it to zirconium chloride and then through magnesium reduction of chloride [1]. The top 3 zirconium reserve holding countries are Australia, South Africa, and Ukraine while the top 3 producers are Australia, South Africa, and China [1].
History
Origin of its Name: The element is named from the Arabic word ‘zargun’, which means ‘gold-colored’ [1].
Who discovered it: Zirconium was discovered by the German chemist Martin Heinrich Klaproth [1].
When and How was it Discovered
In the ancient times, gemstones containing zirconium were known as zircon [1]. Martin Klaproth, in 1789, investigated zircon and separated the new element in the form of baddeleyite or zirconia, the oxide of zirconium [1].
Klaproth could not successfully isolate the pure metal while Humphry Davy was also unsuccessful in splitting zirconia through electrolysis in 1808 [1, 5]. In 1824, the Swedish chemist Jöns Jacob Berzelius isolated zirconium in black, powdery form by heating potassium hexafluorozirconate with potassium [1].
However, the entirely pure form was not produced until 1925 when the Dutch chemists Jan Hendrik de Boer and Anton Eduard van Arkel decomposed zirconium tetraiodide (ZrI4) [1]. Today, the metal is manufactured in bulk through reduction of ZrI4 by magnesium [1].
Zirconium Identification |
|||
| Atomic number | 40 [1] | ||
| CAS number | 7440-67-7 [1] | ||
| Position in the periodic table | Group | Period | Block |
| 4 [1] | 5 [1] | d [1] | |
Properties and Characteristics of Zirconium
General Properties |
||||||||||||||||
| Relative atomic mass | 91.224 [1] | |||||||||||||||
| Atomic mass | 91.224 amu [1] | |||||||||||||||
| Molar mass | 91.2240 g/mol [6] | |||||||||||||||
Physical Properties |
||||||||||||||||
| Color | Silvery, grayish-white [1, 5] | |||||||||||||||
| Melting point/freezing point | 1854 °C, 3369 °F [1] | |||||||||||||||
| Boiling point | 4406 °C, 7963 °F [1] | |||||||||||||||
| Density | 6.52 g cm-3 [1] | |||||||||||||||
| State of matter at room temperature (solid/liquid/gas) | Solid [1, 5] | |||||||||||||||
| Hardness | ||||||||||||||||
| – Brinell | 650 MPa [7] | |||||||||||||||
| – Mohs | 5 [7] | |||||||||||||||
| – Vickers | 904 MPa [7] | |||||||||||||||
| Electrical Conductivity | 2.4 X 106 S/m [7] | |||||||||||||||
| Charge | +4 [8] | |||||||||||||||
| Thermal (heat) conductivity | 23 W/(m K) [7] | |||||||||||||||
| Specific heat | 278 J kg-1 K-1 [1] | |||||||||||||||
| Bulk modulus | Unknown [1] | |||||||||||||||
| Shear modulus | Unknown [1] | |||||||||||||||
| Young’s modulus | Unknown [1] | |||||||||||||||
| Vapor pressure | ||||||||||||||||
| – Temperature (K) | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 | 2200 | 2400 | |||||
| – Pressure (Pa) | – | – | – | – | – | 1.05 X 10-10 | 6.17 X 10-8 | 8.68 X 10-6 | 0.45 X 10-3 | 0.011 | 0.155 | |||||
Chemical Properties |
||||||||||||||||
| Oxidation state/Oxidation number | +1, +2, +3, +4 [1] | |||||||||||||||
| Isotopes | Isotope | Mass | Abundance (%) | Half-life | Mode of decay | |||||||||||
| 90Zr | 89.905 | 51.45 | – | – | ||||||||||||
| 91Zr | 90.906 | 11.22 | – | – | ||||||||||||
| 92Zr | 91.905 | 17.15 | – | – | ||||||||||||
| 94Zr | 93.906 | 17.38 | > 1017 y | β-β- | ||||||||||||
| 96Zr | 95.908 | 2.8 | 2.3 X 1019 y | β-β- | ||||||||||||
| > 1.7 X 1018 y | β- | |||||||||||||||
Atomic Data of Zirconium (Element 40)
| Valence electrons | 4 [9] | |||||||
| Quantum numbers | ||||||||
| – n | 4 [10] | |||||||
| – ℓ | 2 [10] | |||||||
| – mℓ | -1 [10] | |||||||
| – ms | +1/2 [10] | |||||||
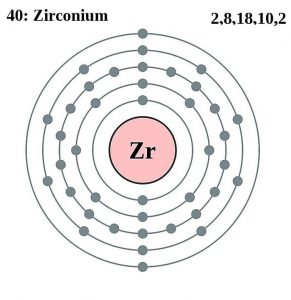
| Electron configuration (noble gas configuration) | [Kr] 4d25s2 [1] | |||||||
| Atomic structure | ||||||||
| – Number of electrons | 40 [5] | |||||||
| – Number of neutrons | 51 [5] | |||||||
| – Number of protons | 40 [5] | |||||||
| Radius of Atom | ||||||||
| – Atomic radius | 2.23 Å [1] | |||||||
| – Covalent radius | 1.64 Å [1] | |||||||
| Electronegativity (Pauling-scale) | 1.33 [1] | |||||||
| Electron affinity | 41.103 [1] | |||||||
| Ionization energy (kJ mol-1) | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
| 640.074 | 1264 | 2218.2 | 3313.31 | 7752.404 | – | – | – | |
Zirconium Electron Configuration (Bohr Model)
What is it Used for
- Since it is not good at absorbing neutrons, it is used to provide the outer covering for the fuel rods in nuclear reactors [1, 5]. The reactors in nuclear power stations can have over 100,000 meters of zirconium tubing [1].
- Being superconductive at low temperatures, the alloy of zirconium and niobium is used for making superconducting magnets [1, 5].
- Zirconium(IV) oxide is used to make ultra-strong ceramics, which can be used for manufacturing crucibles, furnace linings, abrasives, and foundry bricks because of its heat-resistant properties [1]. It is used in antiperspirants, cosmetics, microwave filters, and food packaging industries [1].
- The natural gemstone zircon is available in various colors, out of which the most desirable is the golden one that is used for making jewelry [1]. The colorless, cubic zirconia is used as a synthetic gemstone, and when cut, it resembles a diamond [1].
- Since zirconium is exceptionally resistant to corrosion, it is useful in corrosive environments, and its alloys are found in heat exchangers, pipes, and fittings [5].
- Zirconium silicate mixed with praseodymium or vanadium produces yellow and blue pigments for painting pottery [1].
- Porcelain veneered zirconium crowns, implant dentures, and bridges are used for replacing lost teeth [11].
- Since zirconium powder can naturally ignite in air, it may be used in explosive devices [2].
Zirconium Toxicity
Zirconium is known to have low toxicity, and if exposed, people may experience irritation of skin and eyes [12]. If inhaled, it can cause the formation of tumors in the lungs [12]. It is not known to play any biological role [1].
Interesting Facts
- It is reported that over 1.5 million tons of zircon are mined annually, mainly in South Africa and Australia [1].
- Zirconium is graphically represented as a scarab beetle because zircon gemstones were used in jewelry by the ancient Egyptians who related scarab beetles with creation, transformation, and regeneration [1].
- The element is so strong that it can be used for producing knives and scissors [1].
Zirconium Cost
The price of pure zirconium is about $1.57 per gram, and in bulk, it costs roughly $0.16 per gram [5].
- References
- http://www.rsc.org/periodic-table/element/40/zirconium
- https://www.livescience.com/34610-zirconium.html
- https://education.jlab.org/itselemental/ele040.html
- https://education.jlab.org/itselemental/iso040.html
- https://www.chemicool.com/elements/zirconium.html
- https://www.webqc.org/molecular-weight-of-zirconium.html
- http://periodictable.com/Elements/040/data.html
- https://www.chemthes.com/entity_datapage.php?id=857
- https://zirconium2015.weebly.com/about-zirconium.html
- http://chemistry-reference.com/q_elements.asp?Symbol=Zr
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3399177/
- http://metalpedia.asianmetal.com/metal/zirconium/health.shtml