Dysprosium
Dysprosium (pronunciation: dis-PROH-zee-em) is a silvery rare earth metal classified as a lanthanide and represented by the atomic symbol Dy [1, 2]. It is composed of seven naturally occurring isotopes, out of which 164Dy is the most naturally abundant (with 28% abundance) [1]. Moreover, 29 radioactive isotopes have been produced, the most stable one being 154Dy, characterized by a half-life of 3 million years [3].
Where is Dysprosium Found
Dysprosium, like many other lanthanides, is extracted from the minerals bastnaesite and monazite, as well as from some other minerals like fergusonite and xenotime [1]. It can be obtained from these minerals through the process of solvent extraction and ion exchange [1]. It can as well be manufactured by reducing dysprosium trifluoride using calcium metal [1]. The top 3 dysprosium reserve-holding nations are China, CIS Countries, and the USA whereas the top 3 dysprosium producers include China, Russia, and Malaysia [1].
History
Origin of its Name: The name has been derived from the Greek word ‘dysprositos,’ which means hard to get [1].
Who discovered it: The French chemist Paul-Emile Lecoq de Boisbaudran discovered Dysprosium [1, 4].
When and How was Dysprosium Discovered
In 1886, the element dysprosium was discovered by de Boisbaudran who carried out a series of research on the compound yttrium oxide involving endless precipitations on a marble slab of the fireplace at his home in Paris [1]. The compound was first produced in 1794, and since then several lanthanoids have been extracted, including erbium in 1843, holmium in 1878, and then dysprosium [1].
In 1950, pure samples of the element were first isolated using the ion-exchange chromatography technique, which was developed by the Canadian-American chemist Frank Spedding and his co-workers at the Iowa State University [1]. Today, that separation technique has been replaced by the liquid-liquid exchange technology [1].
Dysprosium Identification |
|||
| Atomic number | 66 [1, 6] | ||
| CAS number | 7429-91-6 [1] | ||
| Position in the periodic table | Group | Period | Block |
| Lanthanides [1] | 6 [1] | f [1] | |
Properties and Characteristics of Dysprosium
General Properties |
||||||||||||||||
| Relative atomic mass | 162.500 [1] | |||||||||||||||
Physical Properties |
||||||||||||||||
| Color | Bright silvery-white [1, 4] | |||||||||||||||
| Melting point/freezing point | 1412 °C, 2574 °F [1] | |||||||||||||||
| Boiling point | 2567 °C, 4653 °F [1] | |||||||||||||||
| Density | 8.55 g cm-1 [1] | |||||||||||||||
| State of matter at room temperature (solid/liquid/gas) | Solid [1, 4] | |||||||||||||||
| Hardness | ||||||||||||||||
| – Brinell | 500 MPa [5] | |||||||||||||||
| – Mohs | 1.8 [5] | |||||||||||||||
| – Vickers | 540 MPa [5] | |||||||||||||||
| Electrical conductivity | 1.1X106 S/m [5] | |||||||||||||||
| Thermal (heat) conductivity | 11 W/(m.K) [5] | |||||||||||||||
| Specific heat | 173 J kg-1 K-1 [1] | |||||||||||||||
| Bulk modulus | 40.5 GPa [1] | |||||||||||||||
| Shear modulus | 24.7 GPa [1] | |||||||||||||||
| Young’s modulus | 61.4 GPa [1] | |||||||||||||||
| Vapor pressure | ||||||||||||||||
| – Temperature (K) | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 | 2200 | 2400 | |||||
| – Pressure (Pa) | – | – | 1.54X 10-8 | 8.21X 10-5 | 2.41X 10-2 | 1.362 | 27.5 | – | – | – | – | |||||
Chemical Properties |
||||||||||||||||
| Oxidation state/Oxidation number | +2 +3 (+4) [1] | |||||||||||||||
| Isotopes | Isotope | Mass | Abundance | Half-life | Mode of decay | |||||||||||
| 156Dy | 155.924 | 0.056 | – | – | ||||||||||||
| 158Dy | 157.924 | 0.095 | – | – | ||||||||||||
| 160Dy | 159.925 | 2.32900 | – | – | ||||||||||||
| 161Dy | 160.927 | 18.889 | – | – | ||||||||||||
| 162Dy | 161.927 | 25.475 | – | – | ||||||||||||
| 163Dy | 162.929 | 24.896 | – | – | ||||||||||||
| 164Dy | 163.929 | 28.26 | – | – | ||||||||||||
Atomic Data of Dysprosium (Element 66)
| Valence electrons | 3 [5] | |||||||
| Quantum numbers | 5I8 [5] | |||||||
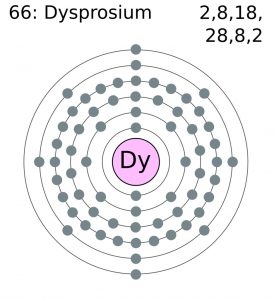
| Electron configuration (noble gas configuration) | [Xe] 4f106s2 [1] | |||||||
| Atomic structure | ||||||||
| – Number of electrons | 66 [7] | |||||||
| – Number of neutrons | 97 [7] | |||||||
| – Number of protons | 66 [7] | |||||||
| Radius of Atom | ||||||||
| – Atomic radius | 2.31 Å [1] | |||||||
| – Covalent radius | 1.80 Å [1] | |||||||
| Electronegativity (Pauling-scale) | 1.22 [1] | |||||||
| Electron affinity | Unknown [1] | |||||||
| Ionization energy (kJ mol-1) | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
| 573.017 | 1125.98 | 2199.9 | 4001.25 | – | – | – | – | |
Dysprosium Bohr Model
What is Dysprosium used for
Since it reacts with air and water, dysprosium does not have much use in its pure metallic form [1]. Its alloys and compounds have the following applications:
- Dysprosium alloys are used for producing neodymium-based magnets because they have better resistance to demagnetization at elevated temperatures. Therefore, these magnets are used in generators, motors, electrical vehicles, and wind turbines [1].
- Compounds of dysprosium like dysprosium halide are applied in commercial lighting, including halide discharge lamps, helping in producing an intense white light [1].
- A mixture containing nickel and dysprosium oxide is utilized in making the control rods used in nuclear reactors to absorb neutrons for a long period without contracting or expanding [1].
Dysprosium Health Effects and Hazards
While soluble salts of dysprosium can be mildly toxic when ingested, animal studies have indicated that taking a very high amount of dysprosium (dose of 500 g) can be life-threatening for humans [8].
Interesting Facts about Dysprosium
- Since Dysprosium does not have a hard texture, it can be easily cut with a knife [9].
- The element 66 has been visually depicted by an image of a nuclear reactor to reflect its use in the control rods of a nuclear reactor [1].
Dysprosium Price
The pure form of Dysprosium costs about $450-$500 per 100g, but in bulk, it costs approximately $30-$40 per 100g [4].
- References
- http://www.rsc.org/periodic-table/element/66/dysprosium
- https://www.livescience.com/38292-dysprosium.html
- https://www.thoughtco.com/dysprosium-facts-element-66-or-dy-4125571
- https://www.chemicool.com/elements/dysprosium.html
- http://periodictable.com/Elements/066/data.html
- https://education.jlab.org/itselemental/ele066.html
- http://www.chemicalelements.com/elements/dy.html
- https://www.lenntech.com/periodic/elements/dy.htm
- https://factfile.org/10-facts-about-dysprosium