Holmium
What is Holmium
Holmium (pronunciation: HOHL-mee-em) is a silvery metal belonging to the group of lanthanides and represented by the chemical symbol Ho [1, 2]. It has one stable naturally occurring isotope, 165Ho, with a natural abundance of 100% while its most stable radioisotope, 163Ho has a half-life period of 4,570 years [3].
Where is Holmium Found
Although Holmium does not freely occur in nature, it is found as a component in several minerals including bastnaesite and monazite [1, 4]. Commercially, it is obtained from monazite, bastnaesite, and xenotime ores through ion exchange and is isolated by calcium reduction of its anhydrous fluoride [1, 4, 5]. While top 3 holmium producing countries include China, Russia, and Malaysia, the top 3 nations with the largest holmium holdings in the world are China, the CIS countries, and the USA [1].
History
Origin of its Name: It is derived from ‘Holmia’, the Latin name for the Swedish capital Stockholm [1].
Who discovered it: Holmium was discovered by the Swedish chemist Per Teodor Cleve, and independently by the Swiss chemist Louis Soret along with Marc Delafontaine [1].
When and How was it Discovered
In 1878, Louis Soret and Marc Delafontaine while investigating yttrium at Geneva found that it was contaminated with other lanthanoids, observing unexplained spectral lines [1, 4]. They extracted a new element ‘X’, which is now known as holmium [4]. In that same year, Per Teodor Cleve worked with erbium at Uppsala to discover a brown and a green substance, which he named holmia (holmium oxide) and thulia (thulium oxide) respectively [4].
The pure form of holmium was separated by the Swedish botanist Otto Holmberg [4].
Holmium Identification |
|||
| Atomic number | 67 [1] | ||
| CAS number | 7440-60-0 [1] | ||
| Position in the periodic table | Group | Period | Block |
| Lanthanides [1] | 6 [1] | f [1] | |
Properties and Characteristics of Holmium
General Properties |
||||||||||||||||
| Atomic mass | 164.930 amu [1] | |||||||||||||||
| Relative atomic mass | 164.930 [1] | |||||||||||||||
Physical Properties |
||||||||||||||||
| Color | Bright silvery-white [1, 4] | |||||||||||||||
| Melting point/freezing point | 1472 °C, 2682 °F [1] | |||||||||||||||
| Boiling point | 2700 °C, 4892 °F [1] | |||||||||||||||
| Density | 8.80 g cm-3 [1] | |||||||||||||||
| State of matter at room temperature (solid/liquid/gas) | Solid [1, 4] | |||||||||||||||
| Hardness | ||||||||||||||||
| – Brinell | 746 MPa [6] | |||||||||||||||
| – Mohs | 1.65 [6] | |||||||||||||||
| – Vickers | 481 MPa [6] | |||||||||||||||
| Electrical conductivity | 1.1X106 S/m [6] | |||||||||||||||
| Thermal (heat) conductivity | 16 W/(m K) [6] | |||||||||||||||
| Specific heat | 165 J kg-1 K-1 [1] | |||||||||||||||
| Bulk modulus | 40.2 GPa [1] | |||||||||||||||
| Shear modulus | 26.3 GPa [1] | |||||||||||||||
| Young’s modulus | 64.8 GPa [1] | |||||||||||||||
| Vapor pressure | ||||||||||||||||
| – Temperature (K) | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 | 2200 | 2400 | |||||
| – Pressure (Pa) | – | – | 3.20 X 10-9 | 2.32 X 10-5 | 8.37 X 10-3 | 0.546 | 12.3 | – | – | – | – | |||||
Chemical Properties |
||||||||||||||||
| Oxidation state/Oxidation number | (+2) +3 [1] | |||||||||||||||
| Isotopes | Isotope | Mass | Abundance (%) | Half-life | Mode of decay | |||||||||||
| 165Ho | 164.930 | 100 | – | – | ||||||||||||
Atomic Data of Holmium (Element 67)
| Valence electrons | 2 [7] | |||||||
| Quantum numbers | ||||||||
| – n | 4 [7] | |||||||
| – ℓ | 3 [7] | |||||||
| – mℓ | 0 [7] | |||||||
| – ms | -1/2 [7] | |||||||
| Electron configuration (noble gas configuration) | [Xe] 4f116s2 [1] | |||||||
| Atomic structure | ||||||||
| – Number of electrons | 67 [4] | |||||||
| – Number of neutrons | 98 [4] | |||||||
| – Number of protons | 67 [4] | |||||||
| Radius of Atom | ||||||||
| – Atomic radius | 2.30 Å [1] | |||||||
| – Covalent radius | 1.79 Å [1] | |||||||
| Electronegativity (Pauling-scale) | 1.23 [1] | |||||||
| Electron affinity | Unknown [1] | |||||||
| Ionization energy (kJ mol-1) | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
| 580.987 | 1138.5 | 2203.73 | 4100.6 | – | – | – | – | |
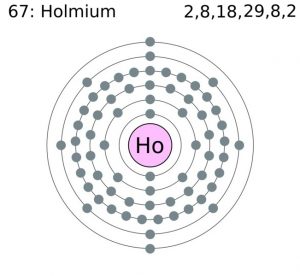
Holmium Electron Configuration (Bohr Model)
How is Holmium Used
- Since it is an excellent absorber of neutrons, its isotopes are used in the control rods of nuclear reactors to control fission chain reactions [1, 4].
- Holmium has special magnetic properties, and so its alloys are used for producing magnets and flux concentrators for high-intensity magnetic fields [4].
- Holmium-doped solid-state lasers are used for medical procedures including breaking-up of kidney stones (lithotripsy), and benign prostatic hyperplasia surgeries like prostate enucleation (HoLEP), resection (HoLRP), and ablation (HoLAP) [4, 8].
- Holmium oxide or holmia can be used as a red or yellow coloring agent for cubic zirconia and glass [4].
Holmium Toxicity
The element 67 is not considered to be harmful because it has low toxicity [4]. However, holmium salts can be toxic if orally consumed, inhaled, or injected in large amounts [9].
Interesting Facts
- Holmium is graphically represented by an image of the coat of arms of Stockholm, the Swedish capital after which the element is named [1].
- The element quickly oxidizes at high temperatures and in the moist air [5].
Cost of Holmium
The cost of pure holmium is $8.6 per gram and $8600 per kg [4], but in bulk, its price is $10 per gram [10].
- References
- http://www.rsc.org/periodic-table/element/67/holmium
- https://education.jlab.org/itselemental/ele067.html
- https://www.webelements.com/holmium/isotopes.html
- https://www.chemicool.com/elements/holmium.html
- https://www.livescience.com/38360-facts-about-holmium.html
- http://www.periodictable.com/Elements/067/data.html
- http://chemistry-reference.com/q_elements.asp?Symbol=Ho&language=en
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4526611/
- http://nautilus.fis.uc.pt/st2.5/scenes-e/elem/e06740.html
- http://hobart.k12.in.us/ksms/PeriodicTable/holmium.htm








