Aufbau Principle
An atom consists of a nucleus around which electrons revolve in well-defined orbits. The orbits are characterized by sublevels or subshells, which are organized according to their increasing energies.
What is Aufbau Principle
The construction of a model of an atom begins by filling up the electrons in the lowest energy subshell. Aufbau principle gives a model for filling the electrons in the empty subshells of an atom in its ground state. According to this principle, electrons first fill up the lower energy subshells before progressively filling up the higher ones [1-4].
‘Aufbau’ comes from the German word Aufbauen, which means ‘building-up’. In other words, the atom builds up as electrons occupy the various subshells. Several scientists have contributed to the development of the Aufbau principle. Danish physicist Niels Bohr was the first to propose a quantum mechanics model of the atom in the 1920s.
Filling Up of Electrons in Subshells
Quantum numbers are used to designate the state of an electron. There are four quantum numbers, of which two are concerned with the subshells. They are principal quantum number n and azimuthal quantum number l [1-3,5].
The sum of n and l determines the order of increasing energies of the subshells. The electrons fill up the subshell according to the increasing value of (n+l). n and l take discrete values, like n = 0, 1, 2, … and l = 0, 1, 2, and 3. For example, n = 3 and l = 1 refer to the 3p subshell.
If two subshells have the same (n+l) value, the electron will occupy the lower n subshell. For example, both 3p and 4s have (n+l) value that is equal to 4. Since 3p has a lower n value, the electron will occupy 3p and not 4s.
Based on this information, the following table gives the order of filling the subshells.
| Subshell | Principle quantum number (n) | Azimuthal quantum number (l) | Sum (n + l) |
|---|---|---|---|
| 1s | 1 | 0 | 1 |
| 2s | 2 | 0 | 2 |
| 2p | 2 | 1 | 3 |
| 3s | 3 | 0 | 3 |
| 3p | 3 | 1 | 4 |
| 4s | 4 | 0 | 4 |
| 3d | 3 | 2 | 5 |
| 4p | 4 | 1 | 5 |
| 5s | 5 | 0 | 5 |
| 4d | 4 | 2 | 6 |
| 5p | 5 | 1 | 6 |
| 6s | 6 | 0 | 6 |
| 4f | 4 | 3 | 7 |
| 5d | 5 | 2 | 7 |
| 7s | 7 | 0 | 7 |
| 5f | 5 | 3 | 8 |
| 6d | 6 | 2 | 8 |
| 7p | 7 | 1 | 8 |
| 8 | 8 | 0 | 8 |
| … | … | … | … |
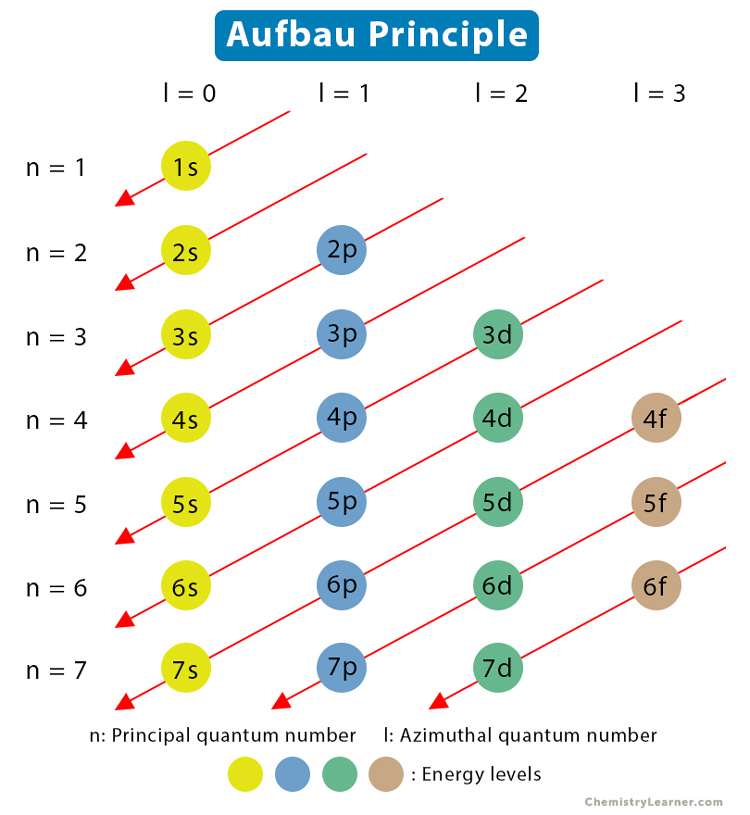
How to Construct Aufbau Diagram
According to the Aufbau principle, the order of filling the subshells increases with (n + l) values. Hence, this information can be represented by a diagram known as the Aufbau diagram (see image below). The following points are observed to construct the diagram [1, 2, 5].
- n values are written in a column on the left. l values are written in a row on the top.
- n and l values combine to represent the subshells.
- Lines joining the subshells having the same (n + l) value are drawn diagonally.
- Arrows are included that point toward increasing n on the diagonals.
Aufbau Principle and Electron Configuration
The Aufbau diagram combined with the number of electrons that occupy each subshell can determine the electron configuration of many elements [1, 2, 7].
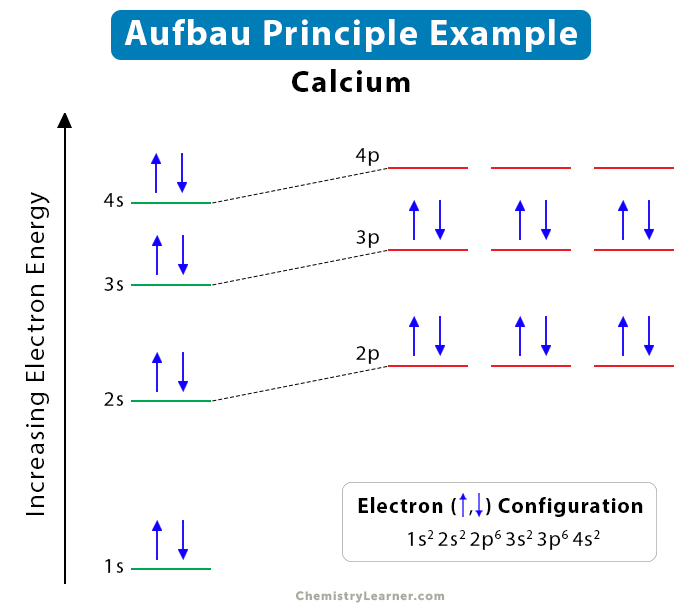
Example of Calcium Atom
Let us take the example of the calcium atom in order to illustrate this concept step by step. The atomic number of calcium is 20. It means that the calcium atom has 20 protons and 20 electrons. The following steps indicate how these electrons fill the subshells.
Step 1: The lowest energy subshell is 1s with a maximum occupancy of 2 electrons. Therefore, the first 2 electrons will occupy 1s.
Step 2: The next higher energy subshell is 2s with a maximum occupancy of 2. Therefore, the next two electrons will occupy 2s.
Step 3: The third highest energy level is 2p which has a maximum occupancy of 6 electrons. Therefore, the next 6 electrons will occupy 2p.
Step 4: The fourth highest energy level is 3s with a maximum occupancy of 2 electrons. Therefore, the next 2 electrons will occupy 3s.
Step 5: The fifth highest energy level is 3p with a maximum occupancy of 6 electrons. Therefore, the next 6 electrons will occupy 3p.
Step 6: Only 2 electrons are remain. The sixth highest energy level is 4s, with a maximum occupancy of 2 electrons. Therefore, the last 2 electrons will occupy 4s.
Hence, the electron configuration of calcium is given by,
1s2 2s2 2p6 3s2 3p6 4s2
The image below summarizes the above steps.
The following table shows the electron configuration of some common elements.
| Element | Atomic Number | Electron Configuration |
|---|---|---|
| Carbon | 6 | 1s2 2s2 2p2 |
| Oxygen | 8 | 1s2 2s2 2p4 |
| Neon | 10 | 1s2 2s2 2p6 |
| Magnesium | 12 | 1s2 2s2 2p6 3s2 |
| Silicon | 14 | 1s2 2s2 2p6 3s2 3p2 |
| Sulfur | 16 | 1s2 2s2 2p6 3s2 3p4 |
Aufbau Principle Exceptions
The Aufbau principle is not a general principle that applies to all atoms. Spectroscopic observations have shown that certain atoms violate the Aufbau principle [5,6].
1. Transition Metals, Lanthanides, and Actinides
In particular, the transition metals, lanthanides, and actinides have electron configurations that disagree with the Aufbau principle. The following observations have been made.
- In transition metals, the d subshell takes an electron from the s subshell. The exception is palladium, where the 4d subshell takes two electrons from the 5s subshell. This exception is illustrated in the image below.
- In some lanthanides and actinides, the d subshell consumes an electron from the f subshell. The exception is thorium. In thorium, 6d subshell consumes two electrons from 5f.
This exception is attributed to numerous factors, such as the increased stability of half-filled subshells and the relatively low energy difference between the 3d and the 4s subshells.
2. Heavy Nuclei
Another violation of the Aufbau principle is caused by heavy nuclei (Z > 120). As the charge on the nucleus becomes high, the electrons are attracted by large electrostatic forces. When it happens, their velocities are close to the speed of light. In this case, the quantum mechanics approach to determine the electron energies fails.
Limitations of Aufbau Principle
The Aufbau principle assumes that electron energies are fixed for every subshell for all elements. However, in reality, the energy of an electron depends upon the energies of other electrons in the atom [2].
The Aufbau principle cannot predict the electron configuration of atoms on ionization. In other words, it does not explain which electrons are to be removed when an ion is formed from an atom.
For example, the electron configuration of iron (Fe) is 1s2 2s2 2p6 3s2 3p6 4s2 3d6. When iron is ionized to iron (II) or Fe2+, theoretically, its electron configuration should be 1s2 2s2 2p6 3s2 3p6 4s2 3d4. However, experimentally, it has been observed that the electron configuration is 1s2 2s2 2p6 3s2 3p6 3d6. It means that 2 electrons from the 4s subshell are preferentially removed instead of 3d subshell. This phenomenon is commonly observed in many transitional metals. Overall, the Aufbau principle cannot distinguish between two energy levels that are close to each other, like the (n-1)d and ns subshells.






I really love this website . It made me understand a lot of things but please also and some of the things cause I searched something and there was no result like isotopes, etc. In overall I really love this website and I recommend it
Isotopes are related to the nucleus and not to electrons, which is what this article is all about.