Bohrium
What is Bohrium
Bohrium (pronunciation BORE-ee-em [2]), represented by the chemical symbol Bh [1], is a synthetic, radioactive element belonging to the family of transition metals [1, 3]. It has 11 known isotopes of which Bh 270 is most stable with a half-life of about 1 minute [2, 3].
Where Is It Found
It has no natural presence on the earth. A few atoms have been made in laboratories [1].
History
Origin of Its Name: It is named after the Danish atomic physicist, Niels Bohr [1].
Who Discovered It: A group of scientists led by German physicists Peter Armbruster and Gottfried Münzenberg [1, 4].
How Was Bohrium Discovered
In 1975, a team of scientists led by Yuri Oganessian at the Joint Institute of Nuclear Research (JINR) in Dubna, Russia, bombarded bismuth with chromium to make isotope 261 of the element. They published the results in 1976 and submitted a claim for their discovery.
In 1981, the team of Armbruster and Münzenberg at the Geselleschaft für Schwerionenforschung (GSI) in Germany followed the same process to synthesize a single atom of isotope 262. This gave rise to a conflict among the two parties involved and they started negotiating on who discovered it and thereby had the right to name it.
IUPAC decided to attribute the discovery to the GSI team since their submission was more credible though they accepted that JINR was probably the first to produce the metal [1].
Bohrium Identification |
|||
| Atomic Number | 107 [1] | ||
| CAS Number | 54037-14-8 [1] | ||
| Position in the periodic table | Group | Period | Block |
| 7 [1] | 7 [1] | d [1] | |
Properties and Characteristics of Bohrium
General Properties |
|
| Atomic mass | 270 atomic mass units [1] |
| Atomic weight | 270 [1] |
Physical Properties |
|
| Color/appearance | Steel grey (estimated) [3] |
| Malleability | Yes [5] |
| Ductility | Yes [5] |
| Melting point/freezing point | Unknown [1] |
| Boiling point | Unknown [1] |
| Density | Unknown [1] |
| State of matter at room temperature (normal phase) | Solid [1] |
| Hardness | N/A [14] |
| Electrical conductivity | Yes [5] |
Magnetic Properties |
|
| Magnetic ordering | Unknown [6] |
| Magnetic susceptibility | N/A [7] |
Chemical Properties |
|
| Oxidation state/Oxidation number | [+7] [1] |
| Reactivity | Unknown [11] |
Atomic Data of Bohrium (Element 107)
| Valence electrons | 6d5 7s2 [10] (as predicted depending on its position in periodic table) | ||||||
| Quantum numbers | |||||||
| – n | 6 [8] | ||||||
| – ℓ | 2 [8] | ||||||
| – m ℓ | 2 [8] | ||||||
| – m s | +½ [8] | ||||||
| Electron configuration (noble gas configuration) | [Rn] 5f146d57s2 [1] | ||||||
| Crystal structure | Unknown [13] | ||||||
| Atomic structure | |||||||
| – Number of Electrons | 107 [3] | ||||||
| – Number of Neutrons | 163 [3] | ||||||
| – Number of Protons | 107 [3] | ||||||
| Energy levels [3] | |||||||
| – First Energy Level | 2 | ||||||
| – Second Energy Level | 8 | ||||||
| – Third Energy Level | 18 | ||||||
| – Fourth Energy Level | 32 | ||||||
| – Fifth Energy Level | 32 | ||||||
| – Sixth Energy Level | 13 | ||||||
| – Seventh Energy Level | 2 | ||||||
| Radius of atom | |||||||
| – Atomic Radius | Unknown [1] | ||||||
| – Covalent Radius | 1.41 Å [1] | ||||||
| Electronegativity | Unknown [1] | ||||||
| Ionization energy
(kJmol-1) [1] |
1st | 2nd | 3rd | 4th | 5th | 6th | 7th |
| – | – | – | – | – | – | – | |
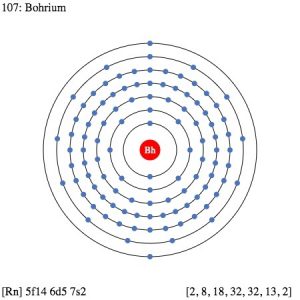
Bohrium Electron Configuration (Bohr Model)
Bohrium Uses
At present, it is used for research purposes only [1].
Is It Dangerous
The highly radioactive property of the metal makes it potentially hazardous [3].
Interesting Facts
- The discoverers wanted to name it as Nielsbohrium (chemical symbol Ns) after Neils Bohr. However, IUPAC suggested the name Bohrium as the first name does not feature in any of the elements named after a person. This principle had a universal acceptance [4].
- It was assigned a placeholder name unnilseptium (chemical symbol Uns) before getting its official IUPAC name [12].
Bohrium (Unnilseptium) Cost
Unknown, as a very small amount has been produced till date [1, 3].
- References
- http://www.rsc.org/periodic-table/element/107/bohrium
- https://education.jlab.org/itselemental/ele107.html
- https://www.chemicool.com/elements/bohrium.html
- https://www.webelements.com/bohrium/
- http://www.elementalmatter.info/element-bohrium.htm
- http://metals.comparenature.com/en/magnetic-properties-of-bohrium/model-69-6
- https://www.schoolmykids.com/learn/interactive-periodic-table/bh-bohrium/
- http://chemistry-reference.com/q_elements.asp?Symbol=Bh&language=en
- http://www.chemistry.patent-invent.com/chemistry/elements/bohrium.html
- http://www.chemistry.patent-invent.com/chemistry/elements/bohrium.html
- https://www.webelements.com/bohrium/chemistry.html
- http://peacecraft.tripod.com/ns.html
- http://www.chemicalelements.com/elements/bh.html
- http://periodictable.com/Properties/A/MohsHardness.an.html







very useful for chemistry students