Bromine
What is Bromine
Bromine (pronounced as BRO-meen) is a volatile, oily liquid halogen with an intermediate reactivity between chlorine and iodine. A diatomic element, it is denoted by the chemical symbol Br [2]. In its gaseous state, also called as bromine gas, it is represented by the chemical formula Br2 [1].
Isotopes
It has 26 isotopes with mass numbers ranging from 68 to 94 of which only Br-79 and Br-81 are the commonly occuring ones, having a stable half-life. [3].
Where is Bromine Found
Br exists naturally in brine deposits found in Israel, USA, and China from where it is extracted through electrolysis. It also occurs in seawater, commonly in the bromide-rich Dead Sea near Israel [1].
History
Origin of its Name: Its name has been derived from the Greek word ‘bromos’, meaning stench [1].
Who Discovered it: Antoine-Jerome Balard and Carl Lowig
When and Where was Bromine Discovered
While evaporating a sample of salty water from Montpellier, France, he found some residue through which chlorine gas was passed. In this process, he extracted some orange-red liquid that was believed to be a new element. In 1826, his discovery details were sent to the French Academy’s journal. However, a year ago, a student from Heidelberg called Carl Lowig had produced the same element from the waters of a natural spring at Keruznach, his hometown. He showed it to his professor, Leopold Gmelin, who asked him to produce more, but since the former got busy with his exams, Balard published his results and became the discoverer [1, 2].
Identification |
|||
| Atomic number | 35 [1] | ||
| CAS number | 7726-95-6 [1] | ||
| Position in the periodic table [1] | Group | Period | Block |
| 17 | 4 | p | |
Classification, Properties and Characteristics of Bromine
General Properties |
|||
| Relative atomic mass | 79.904 [1] | ||
| Atomic mass/weight | 79.904 atomic mass units [7] | ||
| Molar mass (molecular weight) | 192.217 g/mole [3, 8] | ||
| Mass Number | 80 | ||
Physical Properties |
|||
| Color/physical appearance | Deep-red [1] | ||
| Odor | Strong | ||
| Melting point/freezing point | -7.2°C (19°F) [1] | ||
| Boiling point | 58.8°C (137.8°F) [1] | ||
| Density | 3.1028 g/cm3 [1] | ||
| Standard/Natural state at room temperature (solid/liquid/gas) | Liquid [1] | ||
Magnetic Properties [9] |
|||
| Magnetic type | Diamagnetic | ||
| Molar magnetic susceptibility | -7.83×10-10 m3/mol | ||
| Mass magnetic susceptibility | -4.9×10-9 m3/kg | ||
| Volume magnetic susceptibility | -0.0000153 | ||
Chemical Properties |
|||
| Flammability | Non-flammable [10] | ||
| Oxidation state/Oxidation number | −1, +1, +2, +3, +4, +5, +7 [1] | ||
Atomic Data of Bromine (Element 35)
| Valence electrons | 7 [12] | ||||||
| Quantum numbers [11] | |||||||
| – n | 4 | ||||||
| – ℓ | 1 | ||||||
| – mℓ | 0 | ||||||
| – ms | -1/2 | ||||||
| Electron configuration (noble gas configuration) | [Ar] 3d104s24p5[1] | ||||||
| Atomic structure [8] | |||||||
| – Number of Electrons | 35 | ||||||
| – Number of Neutrons | 45 | ||||||
| – Number of Protons | 35 | ||||||
| Radius of atom | |||||||
| – Atomic Radius | 1.85 Å [1] | ||||||
| – Electronegativity | 2.96 [13] | ||||||
| – Covalent Radius | 1.17 Å [1] | ||||||
| Ionization energy [1]
(kJmol-1) |
1st | 2nd | 3rd | 4th | 5th | 6th | 7th |
| 1139.859 | 2083.215 | 3473 | 4563.8 | 5760.2 | 8548.6 | 9938 | |
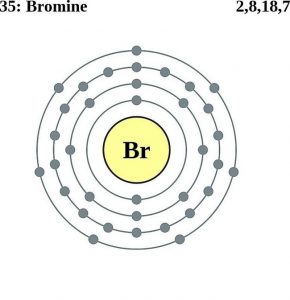
Bromine Atomic Structure (Bohr Model)
What is Bromine Used for
- The element is used in dyes, agricultural products, water sanitizers, insecticides, chemical intermediates, and pharmaceuticals [1, 10].
- It is used as a food additive in brominated vegetable oils, and also as an emulsifier in cold drinks [10].
- Some organic compounds containing bromide and carbon are used in halon fire extinguishers [1].
- Zinc-bromine batteries have been useful in generating power supply to remote areas [14].
Does the Element Have Any Toxic Hazards
Accidental consumption of the liquid halogen or its compounds can lead to nausea and vomiting while inhalation of Br gas could cause respiratory distress, headache, and dizziness. Skin contact with its vapor or liquid is known to cause irritation and burns [6]. In fact, it’s presence as potassium bromate in wheat flours used to make bread have been linked to serious health risks.
Interesting Facts
- 1,2-dibromoethane made from bromine was used as a gasoline additive before unleaded fuels came into use [1].
- An expensive dye called Tyrian purple used during the ancient Roman period was produced by a bromine compound found in murex, a sea mussel [2].
- Bromine gas is heavier than air [6].
Bromine Cost
The price of pure bromine liquid may vary between $5 and $6 per 100 grams.
- References
- http://www.rsc.org/periodic-table/element/35/bromine
- https://education.jlab.org/itselemental/ele035.html
- https://education.jlab.org/itselemental/iso035.html
- https://pubchem.ncbi.nlm.nih.gov/compound/Dibromine#section=Computed-Properties
- https://www.chemicool.com/elements/bromine.html
- https://emergency.cdc.gov/agent/bromine/basics/facts.asp
- http://hobart.k12.in.us/ksms/PeriodicTable/bromine.htm
- https://www.convertunits.com/molarmass/Bromine
- http://periodictable.com/Elements/035/data.html
- https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/316642/Bromine_guidance.pdf
- http://chemistry-reference.com/q_elements.asp?Symbol=Br&language=en
- https://bromine2015.weebly.com/
- https://sciencenotes.org/list-of-electronegativity-values-of-the-elements/
- https://www.sciencedirect.com/science/article/pii/0378775391800597










Year of discovery?
1826