Darmstadtium
What is Darmstadtium
Darmstadtium (pronounced as darm-STADT-ee-em [2]), represented by the chemical symbol Ds [1], is a synthetic, radioactive element belonging to the family of transition metals [3]. It has 15 known isotopes with mass numbers ranging from 267 and 281. The last isotope has the longest half-life of 4 minutes [1].
Where Is It Found
It cannot be found naturally on earth. Small amounts have been synthesized in laboratories by nuclear bombardment [1, 3].
History
Origin of Its Name: It is named after Darmstadt in Germany, the place where it was first produced [1].
Who Discovered It: Czech physicists Sigurd Hofmann, Peter Armbruster and Gottfried Münzenberg [1].
When and Where Was It Discovered: It was discovered on Nov 9, 1994, at the Gesellschaft für Schwerionenforschung (GSI) in Germany [1, 2].
How Was Darmstadtium Discovered
Ds-267 was obtained by Albert Ghiorso and his team at the Lawrence Berkeley National Laboratory, California, by bombarding bismuth with cobalt. However, they could not confirm the results of their experiment at the time. In 1994, Yuri Oganessian and Vladimir Utyonkov made Ds-273 at the Joint Institute of Nuclear Research in Dubna, Russia by bombarding plutonium with sulfur. In the same year, a team at GSI headed by Armbruster and Münzenberg, bombarded lead with nickel to produce isotope 269. The latter group’s findings gained worldwide recognition and were deemed more reliable giving them the authority to name the element [1].
Darmstadtium Identification |
|||
| Atomic Number | 110 [1] | ||
| CAS Number | 54083-77-1 [1] | ||
| Position in the periodic table | Group | Period | Block |
| 10 [1] | 7 [1] | d [1] | |
Properties and Characteristics of Darmstadtium
General Properties |
|
| Atomic mass | 281 atomic mass units [4] |
| Atomic weight | 281 [2] |
Physical Properties |
|
| Color/appearance | Unknown [5] |
| Malleability | Yes [9] |
| Ductility | Yes [9] |
| Melting point/freezing point | Unknown [1] |
| Boiling point | Unknown [1] |
| Density | Unknown [1] |
| State of matter at room temperature (normal phase) | Solid (estimated) [2] |
| Hardness | N/A [5] |
Chemical Properties |
|
| Oxidation state/Oxidation number | [+2], [+4], [+5], [+6] [1] |
| Reactivity | Unknown [11] |
Atomic Data of Darmstadtium (Element 110)
| Valence electrons | 6d9 7s1 [6, 8] | ||||||
| Quantum numbers | |||||||
| – n | 6 [7] | ||||||
| – ℓ | 2 [7] | ||||||
| – m ℓ | 0 [7] | ||||||
| – m s | -½ [7] | ||||||
| Electron configuration (noble gas configuration) | [Rn] 5f146d97s1 [1] | ||||||
| Crystal structure | Bcc (body-centered cubic) [6] | ||||||
| Atomic structure | |||||||
| – Number of Electrons | 110 [3] | ||||||
| – Number of Neutrons | 171 [3] | ||||||
| – Number of Protons | 110 [3] | ||||||
| Energy levels [3] | |||||||
| – First Energy Level | 2 | ||||||
| – Second Energy Level | 8 | ||||||
| – Third Energy Level | 18 | ||||||
| – Fourth Energy Level | 32 | ||||||
| – Fifth Energy Level | 32 | ||||||
| – Sixth Energy Level | 17 | ||||||
| – Seventh Energy Level | 2 | ||||||
| Radius of atom | |||||||
| – Atomic Radius | Unknown [1] | ||||||
| – Covalent Radius | 1.28 Å [1] | ||||||
| Electronegativity | Unknown [1] | ||||||
| Ionization energy
(kJmol-1) [1] |
1st | 2nd | 3rd | 4th | 5th | 6th | 7th |
| – | – | – | – | – | – | – | |
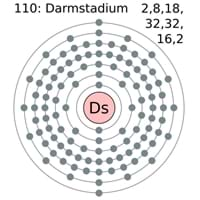
Darmstadtium Electron Configuration (Bohr Model)
Darmstadtium Uses
Since only a few atoms of the element have ever been made, it is only used for research purposes [1].
Can It Be Dangerous
The radioactive metal is unstable and decomposes quickly into other elements that do not pose a health risk [10].
Interesting Facts
- It was known by the name ununnilium (un-one; un-one; nil-zero; ium-the standard prefix for all elements) before getting its official name [9, 12].
- An atomic weight of 281 has earned it the epithet of a super-heavy atom. However, it takes just one-thousandth of a second to decay [9].
Darmstadtium (Ununnilium) Cost
Unknown, as only tiny amounts have been produced in laboratories [1, 3].
- References
- http://www.rsc.org/periodic-table/element/110/darmstadtium
- https://education.jlab.org/itselemental/ele110.html
- https://www.chemicool.com/elements/darmstadtium.html
- http://hobart.k12.in.us/ksms/PeriodicTable/darmstadtium.htm
- https://www.americanelements.com/darmstadtium.html
- https://www.schoolmykids.com/learn/interactive-periodic-table/Ds-Darmstadtium/
- http://chemistry-reference.com/q_elements.asp?Symbol=Ds&language=en
- http://www.chemistry.patent-invent.com/chemistry/elements/darmstadtium.html
- http://www.elementalmatter.info/element-darmstadtium.htm
- https://www.lenntech.com/periodic/elements/ds.htm
- https://www.webelements.com/darmstadtium/chemistry.html
- http://www.actforlibraries.org/an-overview-about-the-chemical-element-darmstadtium/






