Flerovium
What Is Flerovium
Flerovium (pronounced as flee-rove-ee-em) is a radioactive, synthetic metal with the symbol Fl [1]. Though the short half-lives of its five isotopes (mass numbers between 285 and 289) make it difficult to study, its chemical properties (as much could be observed) show it to behave as a metal and a noble gas. That is why this element, along with Copernicium, has been categorized as a volatile metal. Fl belongs to the family of rare earth metals and shows less reactivity [2, 3, 4, 10].
History
Origin of its Name: It is named after the Russian physicist Georgy Flerov, the founder of the Joint Institute for Nuclear Research (JINR) where the element was discovered [1, 4].
Who Discovered it: It was discovered by Yuri Oganessian and Vladimir Utyonkov in 1998.
How was it Discovered
A team led by Oganessian and Utyonkov produced it for the first time at JINR in Dubna, Russia by bombarding plutonium with calcium. The reaction produced a single atom of flerovium-289, the most stable of its isotopes with a half-life of 2.1 seconds [1, 3].
Position of the Element on the Periodic Table [1]
| Group | 14 |
| Period | 7 |
| Block | p |
Properties and Characteristics of Flerovium [1, 2, 3, 4, 5, 7, 9]
General Properties |
||
| Relative atomic mass/ atomic weight | 289 | |
Physical Properties |
||
| Color/appearance | Silvery white or pale gray (estimated) | |
| Odor | Unknown | |
| Melting point/freezing point | Unknown | |
| Boiling point | Unknown | |
| Density | Unknown | |
| State of matter at 200C (normal phase) | Solid (estimated) | |
| Hardness | Unknown | |
| Electrical conductivity | Unknown | |
Magnetic Properties |
||
| Magnetic ordering | Unknown | |
| Magnetic susceptibility | Unknown | |
Chemical Properties |
||
| Oxidation number | Unknown | |
| Oxidation state/Oxidation number | [+2], [+4] | |
Atomic Data of Flerovium [1, 3, 5, 6, 8]
| Atomic number | 114 |
| Valence electrons | 4 |
| Quantum numbers | |
| – n | 7 |
| – ℓ | 1 |
| – m ℓ | 0 |
| – m s | +½ |
| Electron configuration (noble gas configuration) | [Rn] 5f146d107s27p2 |
| Crystal structure | – |
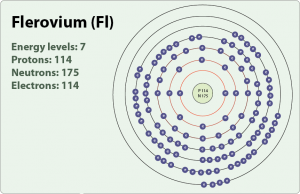
| Atomic structure | |
| – Number of Electrons | 114 |
| – Number of Neutrons | 175 |
| – Number of Protons | 114 |
| Energy levels | |
| – First Energy Level | 2 |
| – Second Energy Level | 8 |
| – Third Energy Level | 18 |
| – Fourth Energy Level | 32 |
| – Fifth Energy Level | 32 |
| – Sixth Energy Level | 18 |
| – Seventh Energy Level | 4 |
| Radius of atom | |
| – Atomic Radius (Å) | Unknown |
| – Covalent Radius (Å) | 1.43 |
| Electronegativity | Unknown |
| Ionization energy
(kJmol-1) |
Unknown |
Flerovium Atomic Structure (Bohr Model)
Flerovium Uses
As the element does not exist naturally and synthesized in small amounts only at the laboratory, it is used for research purposes [3].
Is it Harmful [4]
Being highly radioactive, exposure to it over a long period of time might be dangerous.
Interesting Facts
- It was called Ununquadium (Uuq) until IUPAC named it as flerovium in May, 2012 [4].
- Less than 100 atoms of flerovium have been created to date [7].
- References
- http://www.rsc.org/periodic-table/element/114/flerovium
- https://www.theguardian.com/science/grrlscientist/2013/nov/29/grrlscientist-elements-flerovium-114
- https://education.jlab.org/itselemental/ele114.html
- https://www.chemicool.com/elements/flerovium.html
- http://chemistry-reference.com/q_elements.asp?Symbol=Fl&language=en
- http://periodictable.com/Elements/114/data.html
- http://chemistry.saigon.ro/flerovium.html
- https://periodictable.me/electronegativity-chart/
- https://www.webelements.com/flerovium/compounds.html
- http://hobart.k12.in.us/ksms/PeriodicTable/ununquadium.htm