Heat of Fusion
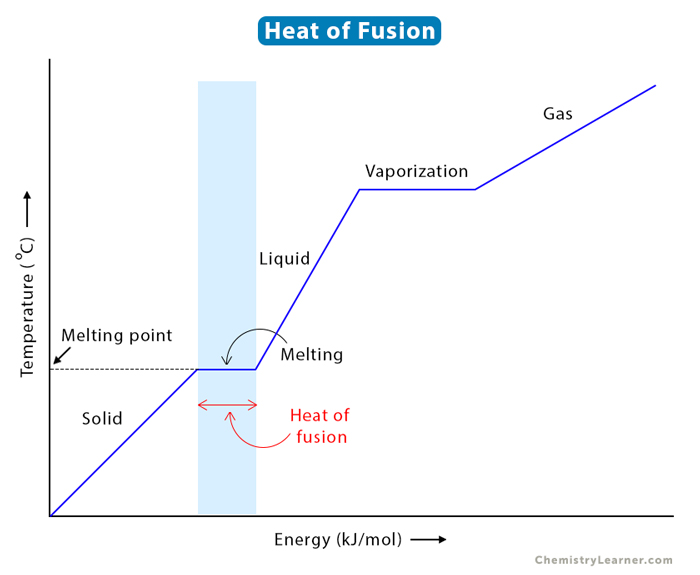
The heat of fusion is the enthalpy change when a unit mass of a substance changes its state from solid to liquid at a constant temperature and pressure. It is sometimes called enthalpy of fusion or latent heat of fusion. The process of going from solid to liquid is known as melting, and the temperature at which it occurs is called the melting point [1-4].
Melting is an endothermic process since the substance absorbs heat during the phase change. A well-known example of melting is ice turning into water.
Molecular Explanation
Behavior at the macroscopic scale dictates that energy must be supplied to a substance to melt. The bonds connecting the atoms or molecules in a crystalline lattice break at the microscopic level. Thus, the molecules are separated and become disordered. Latent heat arises from work done to overcome intermolecular forces. The energy required to melt a solid and the temperature at which it occurs depend on the crystalline structure and the magnitude of the intermolecular forces [1-4].
Equation
Consider m grams of a solid that requires q Joules of energy to change its state completely from solid to liquid. Then, the heat of fusion (ΔHfus) is given by the following formula [1-4]:
ΔHfus = q/m
Symbol: ΔHfus
Unit: Joules/gram (J/g), kiloJoules/kilogram (kJ/kg), or calories/gram (cal/g)
Molar Heat of Fusion
The change in enthalpy when one mole of a substance undergoes a phase change from solid to liquid is called the molar heat of fusion or molar enthalpy of fusion. Let us calculate the molar heat of fusion for water from the heat of fusion [5].
Molar Heat of Fusion for Water
Heat of fusion = 333.55 kJ/kg
Molar mass of water = 18.015 g or 18.015 x 10-3 kg/mol
Molar heat of fusion = 333.55 kJ/1 kg x 18.015 x 10-3 kg/mol = 6 kJ/mol
H2O (s, ice) → H2O (l, water) ΔHfus= 6 kJ/mol
The following table gives the molar heat of fusion of various substances [1].
| Substance | Formula | Melting Point, TM (K) | Molar Heat of Fusion, ΔHfus (kJ/mol) |
|---|---|---|---|
| Neon | Ne | 24 | 0.33 |
| Oxygen | O2 | 54 | 0.44 |
| Methane | CH4 | 90.7 | 0.94 |
| Ethane | C2H6 | 90 | 2.85 |
| Chlorine | Cl2 | 172.2 | 6.4 |
| Carbon tetrachloride | CCl4 | 250 | 2.67 |
| Water | H2O | 273.15 | 6.00 |
| Mercury | Hg | 234 | 2.3 |
| n-Nonane | C9H20 | 353 | 19.3 |
Heat of Solidification
The opposite of melting is freezing or solidification. Here, heat is extracted from the liquid to convert it into a solid. The molecules come closer and become more ordered. The heat liberated by a unit mass of a substance is called heat (enthalpy) of solidification. Solidification is an exothermic process [1-4].
Solved Problems
Problem 1: How much heat is required to melt 10 g of ice at its melting point?
Solution
To calculate the heat (q) required to melt m grams of a liquid, we use the following formula:
q = m ΔHfus
Given, m = 10 g and ΔHfus = 333.55 J/g
q = 10 g x 333.55 J/g = 3335.5 J or 3.34 kJ
Problem 2: What mass of water will be melted at 0 ˚C if 1200 J of heat energy is applied?
Solution
We use the following formula:
q = m ΔHfus
Given, q = 1200 J and ΔHfus = 333.55 J/g
Therefore, m = q/ΔHfus = 1200 J/ 333.55 Jg-1 = 3.6 g
FAQs
Ans. The latent heat of vaporization is higher than fusion because it takes much energy to separate the molecules and vaporize the substance. Once the molecules are entirely separated, the gas expands and occupies ample space.