Seaborgium
What is Seaborgium
Seaborgium (pronounced as see-BOR-ghee-um) is a synthetic radioactive metal, denoted by the symbol Sg, belonging to the family of transuranium elements [1, 6]. Though not much is known about its chemical properties, they are assumed to be similar to tungsten and molybdenum.
Seaborgium Isotopes
It has twelve isotopes of which the most stable one is seaborgium-271, with a half-life of 2.4 minutes. It undergoes decomposition naturally through alpha decay to form rutherfordium-267 [5].
History and Discovery of the Element
It is named after the American scientist and Nobel Prize Winner, Glenn T. Seaborg.
Albert Ghiorso, along with his team of scientists, discovered it in 1974, at the Lawrence Berkley Laboratory, Berkeley, California, US. They bombarded atoms of californium-249 with ions of oxygen-18 with super heavy ion linear accelerator to produce seaborgium-263. This discovery was confirmed in 1993, earning Ghiorso and his team the due credits [2].
Position of the Element on the Periodic Table [1, 2]
| Group | 6 |
| Period | 7 |
| Block | d |
Properties of Seaborgium [1, 2, 3]
General Properties |
||
| Atomic Weight | 271 | |
| Atomic Mass | 266 atomic mass units | |
Physical Properties |
||
| Color/Appearance | A dull to slightly shiny silvery white or metallic gray | |
| Luster | Metallic | |
| Odor | Unknown | |
| Malleability | Unknown | |
| Melting Point/Freezing Point | Unknown | |
| Boiling Point | Unknown | |
| Density | 35 g/cm3 (unconfirmed) | |
| Standard State of Matter at Room Temperature/ Normal Phase (Solid, Liquid, or Gas) | Solid (unconfirmed) | |
| Electrical Conductivity (Sm-1) | Unknown | |
Magnetic Properties |
||
| Magnetic Type | Paramagnetic | |
| Mass Magnetic Susceptibility | 6.66×10-8 m3/Kg | |
| Molar Magnetic Susceptibility | 5.921×10-9 m3/mol | |
| Volume Magnetic Susceptibility | 0.0002978 | |
Chemical Properties |
||
| Flammability | Unknown | |
| Oxidation state/Oxidation number | +6 | |
Atomic Data of the Element [1, 2, 3]
| Atomic Number | 106 | |||
| Valence Electrons | 6d4 7s2 | |||
| Electron Configuration/Noble Gas Configuration | [Rn] 5f14 6d4 7s2 | |||
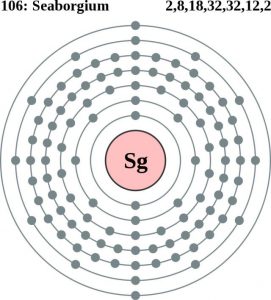
| Atomic Structure | ||||
| – Number of Electrons | 106 | |||
| – Number of Neutrons | 165 | |||
| – Number of Protons | 106 | |||
| Energy Levels | ||||
| – First Energy Level | 2 | |||
| – Second Energy Level | 8 | |||
| – Third Energy Level | 18 | |||
| – Fourth Energy Level | 32 | |||
| – Fifth Energy Level | 32 | |||
| – Sixth Energy Level | 12 | |||
| – Seventh Energy Level | 2 | |||
| Radius of Atom | ||||
| – Atomic Radius | – | |||
| – Covalent Radius | 1.43 Å | |||
| Electronegativity | Unknown | |||
| Ionization Energy | 1st | 2nd | 3rd | 4th |
| – | – | – | – | |
Uses of Seaborgium
As only a few atoms of the element have been synthesized, and it cannot be found freely in nature, it does not have any commercial uses other than in basic scientific studies. However, it may have dangerous effects due to its radioactive nature [1, 3].
Interesting Facts
- Sg is considered to be the heaviest element in group 6 and the third one in the 6d series of transition metals.
- As Seaborgium was named after a living scientist, it led the IUPAC to pass a rule that no element should be named after a living person. However, the ruling was soon removed, keeping einsteinium in mind, another element named after Albert Einstein during his lifetime [4].
- In 2014, some research groups from Mainz and Darmstadt at the RIKEN Nishina Center for Accelerator-based Research (RNC) in Japan established a chemical bond between seaborgium and a carbon atom for the first time [7].
Seaborgium Cost
Since Sg does not exist naturally for its short half-life, it cannot be availed outside of nuclear laboratory production.
- References
- http://www.rsc.org/periodic-table/element/106/seaborgium
- https://education.jlab.org/itselemental/ele106.html
- https://www.chemicool.com/elements/seaborgium.html
- http://www.softschools.com/facts/periodic_table/seaborgium_facts/708/
- https://education.jlab.org/itselemental/iso106.html
- http://www.berkeley.edu/news/berkeleyan/1997/0319/seaborgium.html
- http://www.uni-mainz.de/presse/17619_ENG_HTML.php