Ytterbium
What is Ytterbium
Ytterbium (pronunciation: i-TUR-bee-em) [2] is a malleable, ductile, bright silvery metal [3] that belongs to the group of lanthanides and is represented by the chemical symbol Yb [1]. It has seven stable naturally occurring isotopes, out of which 174Yb has the greatest natural abundance (32.03%), while 176Yb is the most long-lived with a half-life period of 1026 years [1, 4].
Where is it Found
Ytterbium, like other lanthanides, is commonly obtained from the mineral monazite and is separated through solvent extraction and ion exchange procedures [1]. The top 3 ytterbium-producing nations in the world are China, Russia, and Malaysia while the top 3 countries with the largest ytterbium reserves include China, CIS Countries, and the USA [1].
History
Origin of its Name: It is named after Ytterby, a town in Sweden where the element was first found [1].
Who discovered it: Jean Charles Galissard de Marignac, a Swiss chemist, is known for its discovery [1].
When and How was it Discovered
In 1794, the Finnish chemist Johan Gadolin discovered yttrium, a metallic chemical element that the scientists believed was a source of other lanthanoids (rare-earth elements) [1, 2]. In 1843, the Swedish chemist Carl Gustaf Mosander separated yttrium into terbium and erbium [1]. While experimenting with erbium at the University of Geneva in 1878, Charles Marignac was able to isolate ytterbium by heating erbium nitrate, obtaining red erbium oxide and a whitish element that he named ytterbium [1].
In 1937, Klemm and Bonner heated ytterbium chloride together with potassium to produce a small quantity of impure ytterbium metal [1, 5]. A purer form of ytterbium metal was made in 1953 by David Dennison, A. Daane, and Frank Spedding at the Ames Laboratory in Iowa [5].
Ytterbium Identification |
|||
| Atomic number | 70 [1] | ||
| CAS number | 7440-64-4 [1] | ||
| Position in the periodic table | Group | Period | Block |
| Lanthanides [1] | 6 [1] | f [1] | |
Properties and Characteristics of Ytterbium
General Properties |
||||||||||||||||
| Atomic mass | 173.045 amu [11] | |||||||||||||||
| Relative atomic mass | 173.045 [1] | |||||||||||||||
Physical Properties |
||||||||||||||||
| Color | Silvery-white [1, 5] | |||||||||||||||
| Melting point/freezing point | 824 °C, 1515 °F [1] | |||||||||||||||
| Boiling point | 1196 °C, 2185 °F [1] | |||||||||||||||
| Density | 6.90 g cm-3 [1] | |||||||||||||||
| State of matter at room temperature (solid/liquid/gas) | Solid [1, 5] | |||||||||||||||
| Hardness | ||||||||||||||||
| – Brinell | 343 MPa [6] | |||||||||||||||
| – Mohs | Unknown [6] | |||||||||||||||
| – Vickers | 206 MPa [6] | |||||||||||||||
| Electrical conductivity | 0.0351X106 S/m [7] | |||||||||||||||
| Thermal (heat) conductivity | 34.9 W/(m K) [6] | |||||||||||||||
| Specific heat | 155 J kg-1 K-1 [1] | |||||||||||||||
| Bulk modulus | 30.5 GPa [1] | |||||||||||||||
| Shear modulus | 9.9 GPa [1] | |||||||||||||||
| Young’s modulus | 23.9 GPa [1] | |||||||||||||||
| Vapor pressure | ||||||||||||||||
| – Temperature (K) | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 | 2200 | 2400 | |||||
| – Pressure (Pa) | 1.03X 10-9 | 3.84X 10-3 | 6.74 | – | – | – | – | – | – | – | – | |||||
Chemical Properties |
||||||||||||||||
| Oxidation state/Oxidation number | +2 +3 [1] | |||||||||||||||
| Isotopes | Isotope | Mass | Abundance | Half-life | Mode of decay | |||||||||||
| 168Yb | 167.934 | 0.12 | – | – | ||||||||||||
| 170Yb | 169.935 | 2.98 | – | – | ||||||||||||
| 171Yb | 170.936 | 14.09 | – | – | ||||||||||||
| 172Yb | 171.936 | 21.68 | – | – | ||||||||||||
| 173Yb | 172.938 | 16.10 | – | – | ||||||||||||
| 174Yb | 173.939 | 32.03 | – | – | ||||||||||||
| 176Yb | 175.943 | 13.00 | 1026 y | β-β- | ||||||||||||
Atomic Data of Ytterbium (Element 70)
| Valence electrons | 2 [8] | |||||||
| Quantum numbers | ||||||||
| – n | 4 [8] | |||||||
| – ℓ | 3 [8] | |||||||
| – mℓ | 3 [8] | |||||||
| – ms | -1/2 [8] | |||||||
| Electron configuration (noble gas configuration) | [Xe] 4f146s2 [1] | |||||||
| Atomic structure | ||||||||
| – Number of electrons | 70 [5] | |||||||
| – Number of neutrons | 104 [5] | |||||||
| – Number of protons | 70 [5] | |||||||
| Radius of Atom | ||||||||
| – Atomic radius | 2.26 Å [1] | |||||||
| – Covalent radius | 1.78 Å [1] | |||||||
| Electronegativity (Pauling-scale) | Unknown [1] | |||||||
| Electron affinity | -1.93 kJ mol-1 [1] | |||||||
| Ionization energy (kJ mol-1) | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
| 603.435 | 1174.805 | 2416.96 | 4202.9 | – | – | – | – | |
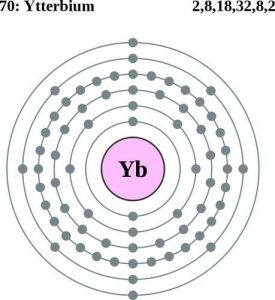
Ytterbium Electron Configuration (Bohr Model)
Uses of Ytterbium
- It can be added to alloys like stainless steel for improving its strength, grain refinement, and other mechanical properties [2, 5].
- Its radioactive isotope 160Yb is used as a source of radiation in portable x-ray imaging equipment [5].
- Ytterbium-doped fiber amplifiers in tunable lasers are used for marking and engraving [5].
- Ytterbium compounds are now increasingly used as industrial catalysts in place of other catalysts that are considered to be hazardous and polluting [1, 5].
- Since the electric resistance of ytterbium increases with increase in physical stress, it is used in strain gauges for seismic stress monitoring [5].
Possible Health Effects
Ytterbium is considered slightly toxic [1], and its compounds can cause skin and eye irritations. Metallic ytterbium dust catches fire spontaneously, and the fumes are harmful to human health [9].
Interesting Facts
- The element 70 is sometimes represented by an image based on ancient rock art found in Sweden [1], because the element was found in the same country.
- Two atomic clocks, based on ytterbium, were combined by scientists at the NIST (National Institute of Standards and Technology) in 2016 to create the most stable clock in the world [10].
Cost of Ytterbium
While 100 grams of pure ytterbium cost around $1,400, the same amount of the element in bulk costs around $550 [11].
- References
- http://www.rsc.org/periodic-table/element/70/ytterbium
- https://education.jlab.org/itselemental/ele070.html
- https://www.livescience.com/38423-ytterbium.html
- https://www.webelements.com/ytterbium/isotopes.html
- https://www.chemicool.com/elements/ytterbium.html
- https://www.webelements.com/ytterbium/physics.html
- https://chemglobe.org/ptoe/_/70.php
- http://chemistry-reference.com/q_elements.asp?Symbol=Yb&language=en
- http://www.elementsdatabase.com/Ytterbium-Yb-70-element/
- https://www.nist.gov/news-events/news/2016/11/nist-debuts-dual-atomic-clock-and-new-stability-record
- http://hobart.k12.in.us/ksms/PeriodicTable/ytterbium.htm








