Erbium
What is Erbium
Erbium (pronunciation UR-bee-em [2]), represented by the chemical symbol Er [1], belongs to the family of rare earth metals [3]. A mixture of six isotopes with mass numbers 162, 164, 166, 167, 168 and 170 is found in naturally occurring erbium.[1, 3]. It reacts with air, water, halogens and acids [9].
Where Is It Found
3.5 milligrams of it can be found in per kilogram of the earth’s crust. It has an oceanic abundance of 8.7 X 10-7 milligrams per liter [2]. It is mainly found in the minerals monazite and bastnaesite [1].
History
Origin of Its Name: It is named after Ytterby, a village in Sweden [1].
Who Discovered It: Swedish chemist Carl Gustav Mosander [1, 2].
How Was Erbium Discovered
In 1843, Mosander separated gadolinite, a mineral discovered in a quarry near Ytterby, into 3 substances that he named yttria, erbia and terbia. He synthesized terbium and erbium from terbia and erbia respectively. It was later found that terbia was terbium oxide (yellow) and erbia was erbium oxide (pink). However, the early samples of the metal contained other rare earths. A pure sample could only be produced in 1934 by heating purified erbium chloride and potassium by the scientists Wilhelm Klemm and Heinrich Bommer [1, 2].
Erbium Identification |
|||
| Atomic Number | 68 [1] | ||
| CAS Number | 7440-52-0 [1] | ||
| Position in the periodic table | Group | Period | Block |
| Lanthanides [1] | 6 [1] | f [1] | |
Properties and Characteristics of Erbium
General Properties |
|
| Atomic mass | 167.259 atomic mass units [1] |
| Atomic weight | 167.259 [1] |
Physical Properties |
|
| Color/appearance | Silvery white [3] |
| Malleability | Yes [11] |
| Ductility | Yes [11] |
| Melting point/freezing point | 1529°C, 2784°F [1] |
| Boiling point | 2868°C, 5194°F [1] |
| Density | 9.07 g cm-3 [1] |
| State of matter at room temperature (normal phase) | Solid [1] |
| Mohs Hardness | 1.97 [7] |
| Electrical conductivity | 1.2 X 106 S/m [7] |
Chemical Properties |
|
| Oxidation state/Oxidation number | (+2) +3 [10] |
Atomic Data of Erbium (Element 68)
| Valence electrons | 2 [8] | ||||||
| Quantum numbers | |||||||
| – n | 4 [8] | ||||||
| – ℓ | 3 [8] | ||||||
| – m ℓ | 1 [8] | ||||||
| – m s | -½ [8] | ||||||
| Electron configuration (noble gas configuration) | [Xe] 4f126s2 [1] | ||||||
| Crystal structure | Hexagonal [10] | ||||||
| Atomic structure | |||||||
| – Number of Electrons | 68 [3] | ||||||
| – Number of Neutrons | 98 [3] | ||||||
| – Number of Protons | 68 [3] | ||||||
| Energy levels [3] | |||||||
| – First Energy Level | 2 | ||||||
| – Second Energy Level | 8 | ||||||
| – Third Energy Level | 18 | ||||||
| – Fourth Energy Level | 30 | ||||||
| – Fifth Energy Level | 8 | ||||||
| – Sixth Energy Level | 2 | ||||||
| Radius of atom | |||||||
| – Atomic Radius | 2.29 Å [1] | ||||||
| – Covalent Radius | 1.77 Å [1] | ||||||
| Ionic charge | +3 [2] | ||||||
| Electronegativity (Pauling scale) | 1.24 [1] | ||||||
| Ionization energy
(kJmol-1) [1] |
1st | 2nd | 3rd | 4th | 5th | 6th | 7th |
| 589.304 | 1151.07 | 2194.08 | 4119.9 | – | – | – | |
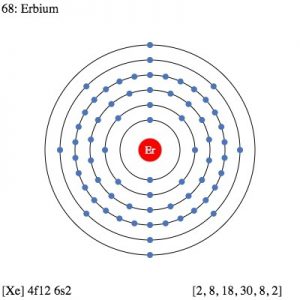
Erbium Electron Configuration (Bohr Model)
Erbium Uses
It has few uses since it slowly tarnishes in air and is decomposed by water [1].
- Being good neutron absorbers, its isotopes are used in nuclear reactor control rods [3].
- It is used in alloys along with Vanadium mostly to decrease the hardness of metals [3].
- It is used in photographic filters as absorbers of infrared light [3].
- It finds application in medical yttrium aluminum garnet (YAG) lasers for skin resurfacing procedures such as acne scar, mole, tattoo and wart removals [3], stretch marks and seborrheic keratosis treatment [6]. The laser is also used in the dental field [5].
- Erbium is doped into the glass fiber of fiber optic cables carrying broadband signals, in order to amplify them [1, 2].
- Erbium 169 with a half-life of 9.4 days is medically used to relieve pain in synovial joints that are related to arthritis [4].
Is It Dangerous
It has a low toxicity [1], though not much is known about its effects on health.
Interesting Fact
- It was one of the first lanthanoid elements to be discovered [1].
Erbium Cost
Pure erbium costs $540 per 100 grams whereas the same amount in bulk costs $65 [3].
- References
- http://www.rsc.org/periodic-table/element/68/erbium
- https://education.jlab.org/itselemental/ele068.html
- https://www.chemicool.com/elements/erbium.html
- https://www.acls-pals-bls.com/nuclear-medicine-and-medical-isotopes/
- https://www.dentistryiq.com/articles/wdj/print/volume-1/issue-4/science/erbium-laser-technology.html
- http://www.jkslms.or.kr/journal/view.html?uid=6&vmd=Full
- http://periodictable.com/Elements/068/data.html
- http://chemistry-reference.com/q_elements.asp?Symbol=Er&language=en
- https://www.webelements.com/erbium/chemistry.html
- https://environmentalchemistry.com/yogi/periodic/Er.html
- http://www.chemistryexplained.com/Di-Fa/Erbium.html









