Neptunium
- What is Neptunium
- Neptunium Isotopes
- Where is Neptunium Found on Earth
- History
- Classification and Position of Neptunium on the Periodic Table [1]
- Properties of Neptunium [1, 2, 3]
- Atomic Properties of the Element [1, 2, 3]
- Neptunium Uses
- Interesting Facts
- Toxicity and Dangerous Effects of Neptunium
- Neptunium Cost
What is Neptunium
Neptunium (pronounced as nep-TOO-nee-em) belongs to the family of actinide elements and is denoted by the chemical symbol Np [1].
Neptunium Isotopes
It has 20 isotopes out of which neptunium-237 is the most stable one with a half-life of 2.14 million years that undergoes alpha decay to form protactinium-233 [3].
Where is Neptunium Found on Earth
The element is known to be present in small quantities associated with uranium ore deposits [1]. Its natural occurrence is due to beta decay of uranium into neptunium-237 and neptunium-239 [3].
History
Origin of its Name: It has been named after the planet Neptune [1].
Who Discovered Neptunium: Edwin McMillan and Philip Abelson were the discoverers of the element [1].
When and Where was it Discovered
In 1934, Enrico Fermi in Italy claimed to be successful in producing elements 93 and 94 by bombarding uranium with neutrons. However, Ida Tacke-Noddack challenged his analysis by pointing out that he actually discovered nuclear fission of uranium. In 1938, similar claims were made by Horia Hulubei and Yvette Cauchoi, but they were rejected, as the new element was not believed at that time to be present in nature.
Finally, in 1940, McMillan and Abelson at Berkeley, California were finally able to confirm the presence of the new element. While bombarding slow neutrons with uranium, beta-rays were produced with unusual properties, suggesting the presence of a new isotope, later confirmed to be the element 93 [1].
Classification and Position of Neptunium on the Periodic Table [1]
| Group Number | Unknown |
| Group Name | Actinides |
| Period | 7 |
| Block | f |
Properties of Neptunium [1, 2, 3]
General Properties |
|||||
| Atomic Mass | 223 atomic mass units | ||||
Physical Properties |
|||||
| Color | Silver-like | ||||
| Luster | Unknown | ||||
| Odor | Unknown | ||||
| Malleability | Unknown | ||||
| Melting point/freezing point | 1191°F (644°C) | ||||
| Boiling Point | 7056°F (3902°C) | ||||
| Density | 20.2 g/cm3 | ||||
| State of matter at room temperature (solid/liquid/gas) | Solid | ||||
| Hardness (Mohs) | Unknown | ||||
| Electrical conductivity | 0.8 x 106 Sm-1 | ||||
| Thermal (heat) conductivity | 6.3 W m-1 K-1 | ||||
Chemical Properties |
|||||
| Flammability | Unknown | ||||
| Oxidation state/Oxidation number | +3, +4, +5, +6, +7 | ||||
Atomic Properties of the Element [1, 2, 3]
| Atomic number | 93 | ||||||
| Valence electrons | 7 | ||||||
| Electron configuration (noble gas configuration) | [Rn] 5f46d17s2 | ||||||
| Atomic structure | |||||||
| – Number of electrons | 93 | ||||||
| – Number of neutrons | 144 | ||||||
| – Number of protons | 93 | ||||||
| Energy levels | |||||||
| – First energy level | 2 | ||||||
| – Second energy level | 8 | ||||||
| – Third energy level | 18 | ||||||
| – Fourth energy level | 32 | ||||||
| – Fifth energy level | 23 | ||||||
| – Sixth energy level | 8 | ||||||
| – Seventh energy level | 2 | ||||||
| Radius of atom | |||||||
| – Atomic radius | 2.39 Å | ||||||
| – Covalent radius | 1.80 Å | ||||||
| Electronegativity | 1.3 | ||||||
| Ionization energy
(kJmol-1) |
1st | 2nd | 3rd | 4th | 5th | 6th | 7th |
| 604.548 | |||||||
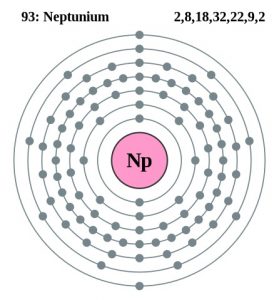
Neptunium Bohr Model
Neptunium Uses
Neptunium-237 is used in neutron detectors. When bombarded with neutrons, it produces plutonium-238 which in turn in is used in spacecraft generators and terrestrial navigation beacons [1, 3].
Interesting Facts
- Neptunium exists in three different physical forms or allotropes; orthorhombic at room temperature, tetragonal above 280°C, and cubic above 577°C [3, 4].
- It is obtained as a byproduct from nuclear reactors as well as spent nuclear fuel rods [1, 4].
Toxicity and Dangerous Effects of Neptunium
Although there is not much information about its hazardous effects, some animal studies have shown its accidental ingestion could cause damage to the liver, kidneys, and brain [5].
Neptunium Cost
It is not available outside of laboratory production.
- References
- http://www.rsc.org/periodic-table/element/93/neptunium
- https://education.jlab.org/itselemental/ele093.html
- https://www.chemicool.com/elements/neptunium.html
- https://www.livescience.com/39808-facts-about-neptunium.html
- https://journals.lww.com/health-physics/Abstract/1963/08000/Acute_Toxicity_of_Neptunium_237_and_Its.4.aspx









Wow!