Protactinium
What is Protactinium
Protactinium is a highly radioactive metal belonging to the family of actinides and denoted by the chemical symbol Pa [1]. It has 27 isotopes with mass numbers ranging from 212 to 238 out of which protactinium-231 is the most stable one with a half-life of 3.276 x 104 years. After which, it forms actinium-277 through alpha decay [3].
History
Origin of its Name: Its name is derived from the Greek word ‘protos’, meaning first, added as a prefix to the element actinium [1].
Who Discovered Protactinium: Kasimir Fajans and Otto Gohring were the discoverers of the element [1].
When and How was it Discovered
In 1900, William Crookes isolated an unidentified radioactive metal from uranium. However, it was only in 1913 when Fajans and Gohring showed that the new element underwent beta-decay and confirmed it to fall in the sequence of elements through uranium decay. It was identified to be isotope protactinium-234 with a half-life of 6 hours and 42 minutes [1].
In 1918, Lise Meitner at the Kaiser Wilhelm Institute, Berlin extracted another isotope protactinium-231 from uranium ore pitchblende [1].
Protactinium Identification |
|||
|
Atomic number |
91 [1] | ||
|
CAS number |
7440-13-3 [1] |
||
|
Position in the periodic table [1] |
Group |
Period |
Block |
|
7 |
f |
||
Properties and Characteristics of Protactinium
General Properties |
||
| Relative atomic mass | 231.036 [1] | |
| Atomic mass | 231.036 atomic mass units [5] | |
Physical Properties |
||
| Color/appearance | Silvery [1] | |
| Luster | Metallic [6] | |
| Melting point/freezing point | 1572°C (2862°F) [1] | |
| Boiling point | 4000°C (7232°F) [1] | |
| Density | 15.4 g/cm3 [1] | |
| State at standard room temperature (solid/liquid/gas) | Solid [1] | |
| Hardness (Brinell, Mohs, Vickers) | Unknown [8] | |
| Thermal conductivity | 47 W m-1K-1 | |
| Electrical conductivity | 5.6 x 106 Sm-1 | |
Chemical Properties |
||
| Oxidation state/Oxidation number | (+2), +3, +4, +5 [1] | |
Atomic Data of Protactinium (Element 91)
| Electron configuration (noble gas configuration) | [Rn] 5f26d17s2 [1] | ||||||
| Crystal Structure | Face-centered cubic (fcc) [3] | ||||||
| Atomic structure [5] | |||||||
| – Number of Electrons | 91 | ||||||
| – Number of Neutrons | 140 | ||||||
| – Number of Protons | 91 | ||||||
| Energy levels [9] | |||||||
| – First energy level | 2 | ||||||
| – Second energy level | 8 | ||||||
| – Third energy level | 18 | ||||||
| – Fourth energy level | 32 | ||||||
| – Fifth energy level | 20 | ||||||
| – Sixth energy level | 9 | ||||||
| – Seventh energy level | 2 | ||||||
| Radius of atom | |||||||
| – Atomic Radius | 2.43 Å [1] | ||||||
| – Covalent Radius | 1.84 Å [1] | ||||||
| Electronegativity | 1.5 [7] | ||||||
| Ionization energy [1]
(kJmol-1) |
1st | 2nd | 3rd | 4th | 5th | 6th | 7th |
| 568.3 | – | – | – | – | – | – | |
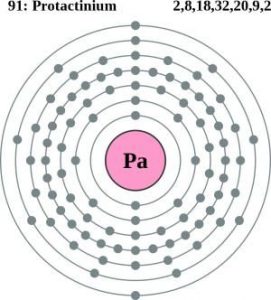
Protactinium Atomic Structure (Bohr Model)
Uses of Protactinium
There are no practical applications of the element outside basic scientific research [1].
Protactinium Hazards
Although the radioactive nature of the element makes it highly toxic, its scarcity in nature does not pose any serious threats [10].
Interesting Facts
- In 1961, 125 g of pure Pa was produced at the Great Britain Atomic Energy Authority by processing 55,000 kg of ore that led to an expenditure of about $500,000 [2].
- Minute amounts of the element exist in uranium ores as well as found in spent fuel rods obtained from nuclear reactors [1].
Protactinium Cost
The price of the metal may vary between $250 to $300 per gram.
- References
- http://www.rsc.org/periodic-table/element/91/protactinium
- https://education.jlab.org/itselemental/ele091.html
- https://www.chemicool.com/elements/protactinium.html
- https://education.jlab.org/itselemental/iso091.html
- http://hobart.k12.in.us/ksms/PeriodicTable/protactinium.htm
- http://www.ivyroses.com/Chemistry/Chemical-Elements_List-ByNameofElement.php
- https://www.nuclear-power.net/protactinium-affinity-electronegativity-ionization/
- http://periodictable.com/Elements/091/data.html
- http://www.chemicalelements.com/elements/pa.html
- https://www.raci.org.au/document/item/470