Cadmium
What is Cadmium
Cadmium (pronounced as KAD-mee-əm) is a naturally-occurring shiny metal denoted by the chemical symbol Cd. Although a non-radioactive element, it is poisonous, causing cancer and prenatal defects, therefore having limited uses. It has eight naturally-existing isotopes of which only Cd-110, Cd-111, and Cd-112 have a stable half-life.
Where is Cadmium Found on Earth
Greenockite and sphalerite are the important natural sources of the metal. On a commercial scale, it is extracted in the vapor form as a by-product of zinc refining.
History
Origin of its Name: It is derived from the Latin word ‘cadmia’ that stands for the mineral calamine.
Who Discovered it: A German chemist called Friedrich Stromeyer
When and Where was Cadmium Discovered
In the early 1800s, zinc oxide was produced by some apothecaries at Hanover in Germany by heating zinc carbonate (cadmia). However, the compound was not pure white but discolored. Later, in 1817 Stromeyer from Gottingen University after a thorough analysis inferred that the discoloration could be due to an unknown element. So he extracted it as a brown oxide and then heated with lampblack to produce a blue-grey metal sample, named as cadmium. While at Halle, Karl Meissner, a German-American physicist, and at Berlin, Karl Karsten, another German scientist worked on the same problem and declared their findings the following year [1].
Identification |
|||
| Atomic number | 48 [1] | ||
| CAS number | 7440-43-9 [1] | ||
| Position in the periodic table [1] | Group | Period | Block |
| 12 | 5 | d | |
Properties and Characteristics of Cadmium
General Properties |
||
| Relative atomic mass | 112.414 [1] | |
| Atomic mass/weight | 112.414 atomic mass units [4] | |
| Molar mass | 112.4110 g/mole [5] | |
Physical Properties |
||
| Color/appearance | Silver with a bluish tinge [1] | |
| Luster | Metallic [6] | |
| Melting point/freezing point | 321.069°C (609.924°F) [1] | |
| Boiling point | 767°C (1413°F) [1] | |
| Density | 8.69 g/cm3 [1] | |
| State of matter at normal room temperature (solid/liquid/gas) | Solid [1] | |
| Specific Heat | 0.230274 J/(g-°C) [7] | |
Chemical Properties |
||
| Oxidation state/Oxidation number | (+1), +2 [1] | |
Atomic Data of Cadmium (Element 48)
| Valence electrons | 1 [8] | ||||||
| Quantum numbers [7] | |||||||
| – n | 4 | ||||||
| – ℓ | 2 | ||||||
| – mℓ | 2 | ||||||
| – ms | -1/2 | ||||||
| Electron configuration (noble gas configuration) | [Kr] 4d105s2 [1] | ||||||
| Atomic structure [4] | |||||||
| – Number of Electrons | 48 | ||||||
| – Number of Neutrons | 64 | ||||||
| – Number of Protons | 48 | ||||||
| Radius of atom | |||||||
| – Atomic Radius | 2.18 Å [1] | ||||||
| – Covalent Radius | 1.40 Å [1] | ||||||
| Ionization energy [1]
(kJmol-1) |
1st | 2nd | 3rd | 4th | 5th | 6th | 7th |
| 867.772 | 1631.404 | 3616.27 | – | – | – | – | |
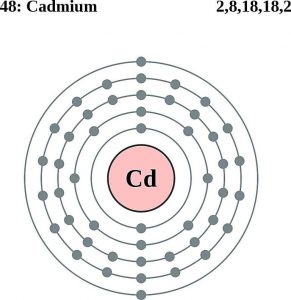
Cadmium Atomic Structure (Bohr Model)
What is Cadmium Used for
- Since cadmium has high corrosion resistance, it’s used in plating bolts, fasteners, and also airplane parts and oil platforms made from stainless steel [1].
- Rechargeable batteries made by combining nickel and cadmium have a longer storage life and low internal resistance [1, 9].
- Helium-cadmium lasers have an excellent beam quality making them useful in spectroscopy, exposing holography, microlithography, and 3-D stereolithography [10].
- As Cd is a good absorber of neutrons, it is used in control rods of nuclear reactors for conducting atomic fission [1].
- Silver alloyed with the element forms low-melting solder used for joining pipes, electrical components, and other metallic equipment [2].
Does it Have Any Toxic Effects
Inhalation of high levels of cadmium dust over a short duration of time can cause several health hazards like lung and kidney damage as a result of poisoning. Also, its accidental ingestion can directly affect the liver and kidneys. [12]
Interesting Facts
- In the past, Cd was used as phosphor coating in cathode ray tubes of color television sets as well as a component in orange, yellow, and red pigments [1].
- It is a soft metal that can be easily cut with a knife [11].
Cadmium Price
The pure form of the metal may cost somewhere between $6 and $7 per gram.
- References
- http://www.rsc.org/periodic-table/element/48/cadmium
- https://education.jlab.org/itselemental/ele048.html
- https://pubchem.ncbi.nlm.nih.gov/compound/cadmium
- http://hobart.k12.in.us/ksms/PeriodicTable/cadmium.htm
- https://www.webqc.org/molecular-weight-of-cadmium.html
- http://webmineral.com/data/Cadmium.shtml#.Wx5FO0iFPIU
- https://www.engineersedge.com/materials/specific_heat_capacity_of_metals_13259.htm
- https://study.com/academy/answer/how-many-valence-electrons-does-cadmium-have.html
- http://www.bodine.com/downloads/docs/Ni-Cad_Battery_Advantages.Article.L0000024.pdf
- https://www.globalspec.com/learnmore/optical_components_optics/lasers/helium_cadmium_hecd_lasers
- https://www.thoughtco.com/cadmium-element-facts-606511
- https://www.osha.gov/SLTC/cadmium/healtheffects.html









