Plutonium
What is Plutonium
Plutonium (pronounced as ploo-TOE-nee-em) is a naturally-occurring radioactive element that belongs to the family of actinide metals. It reacts readily with oxygen, carbon, silicon, nitrogen, and halogens to form stable compounds [7]. Denoted by the chemical symbol Pu, it has 17 isotopes with mass numbers ranging from 227 to 248 of which Pu-224 is the most stable having a half-life of 80.8 million years [1] [3, 4].
Where is Plutonium Found
The main sources of Pu are uranium ores, but most of it is man-made, obtained from irradiated uranium in nuclear reactors through reprocessing. Another method to produce Pu is by reducing plutonium tetrafluoride with calcium [1].
History
Origin of its Name: The name of the element has come from the planet Pluto [1].
Who Discovered it: Glenn Seaborg, Arthur Wahl, Joseph Kennedy, and Edwin McMillan [1]
Discovery Date: December, 1940
When, Where, and How was it Discovered
In December 1940 at Berkeley, California, Seaborg, Wahl, Kennedy, McMillan produced an unknown element by bombarding uranium-238 with deuterium nuclei. It was recognized to be neptunium-238 with a half-life of 2 days that underwent beta decay to form another new element. After a detailed study, its characteristics were found to be similar to uranium [1].
Identification |
|||
| Atomic number | 94 [1] | ||
| CAS number | 7440-07-5 [1] | ||
| Position in the periodic table [1] | Group | Period | Block |
| Actinides | 7 | f | |
Classification, Properties and Characteristics of Plutonium
General Properties |
||
| Relative atomic mass | [244] [1] | |
| Atomic mass/weight | 244 atomic mass units [11] | |
| Molar mass | 238.049 g/mole [12] | |
| Mass Number | 226 | |
Physical Properties |
||
| Color/appearance | Silver [1] | |
| Melting point/freezing point | 640°C (1184°F) [1] | |
| Boiling point | 3228°C (5842°F) [1] | |
| Density | 19.7 g/cm3 [1] | |
| Standard state at room temperature (solid/liquid/gas) | Solid [1] | |
Chemical Properties |
||
| Oxidation state/Oxidation number | +3, +4, +5, +6, +7, (+8) [1] | |
| Flammability | Yes [3] | |
Atomic Data of Plutonium (Element 94)
| Valence electrons | 2 [13] | ||||||
| Quantum numbers [14] | |||||||
| – n | 5 | ||||||
| – ℓ | 3 | ||||||
| – mℓ | 2 | ||||||
| – ms | +1/2 | ||||||
| Electron configuration (noble gas configuration) | [Rn] 5f67s2[1] | ||||||
| Atomic structure [11] | |||||||
| – Number of Electrons | 94 | ||||||
| – Number of Neutrons | 150 | ||||||
| – Number of Protons | 94 | ||||||
| Radius of atom | |||||||
| – Atomic Radius | 2.43 Å [1] | ||||||
| – Covalent Radius | 1.80 Å [1] | ||||||
| Ionization energy [1]
(kJmol-1) |
1st | 2nd | 3rd | 4th | 5th | 6th | 7th |
| 581.421 | 1080.6 | – | – | – | – | – | |
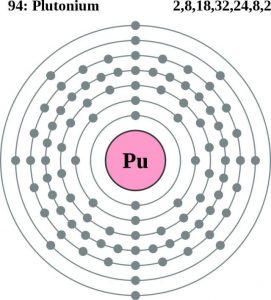
Plutonium Atomic Structure (Bohr Model)
What is Plutonium Used for
- Weapon-grade plutonium, extracted as a byproduct of uranium fuel rods that has been irradiated for two to three months in a reactor, is useful in the military field [5].
- Plutonium-238 pellets with a half-life of 87.7 years is an effective source of electricity for radioactive thermoelectric generators used in space probes [2]. It has also been used to power some cardiac pacemakers [8].
- Pu-239 is used in several nuclear power plant reactors and weapons as a source of energy [2].
- Plutunium-244 is used in some basic scientific studies [9].
Does it Have Any Toxic Effects
Prolonged inhalation of Pu can cause lung cancer, liver cancer, and bone sarcoma as it accumulates in the body and stays for a longer period, making it a lethal poison [3].
Interesting Facts
- Pu turns into a liquid upon reaching its melting point, exhibiting higher-than-normal viscosity, density, and surface tension [6].
- When the element is exposed to air, a coating of plutonium oxide forms on the surface that has pyrophoric properties, causing it to glow in the dark [6]
- Fat man, a massive atomic bomb with a plutonium core, weighing about 6.2 kg, was dropped on Nagasaki during the World War 2 [10].
How Much is the Price of Plutonium
The cost of the pure element may vary between $4000 and $6000 per gram.
SEO Title: Plutonium Facts, Symbol, Discovery, Properties, Uses
SEO Description-