Thulium
What is Thulium
Thulium (pronounced as Thoo-lee-em) [2] is a soft, silver-gray rare earth metal [3] belonging to the Lanthanides group and represented by the symbol Tm [1]. While its most abundant, naturally occurring isotope is 169Tm, it has 34 radioactive isotopes, out of which the most stable one is 171Tm with a half-life period of 1.92 years [4].
Where is it Found
Thulium is found in small quantities in monazite, a mineral that has approximately 20 parts per million (ppm) of the metal in the earth’s crust [1]. It is then commercially extracted through ion-exchange and solvent-extraction processes [1]. The element can as well be isolated through calcium reduction of anhydrous fluoride or by reduction of oxide with lanthanum [1]. While the top 3 thulium producing nations are China, Russia, and Malaysia, the top 3 thulium reserve holders are China, CIS Countries, and Russia [1].
History
Origin of its Name: It is named after Thule, the ancient name that stood for the present-day Scandinavia [1, 2].
Who discovered it: The Swedish chemist Per Teodor Cleve is credited for the discovery of thulium [1].
When and How was it discovered
Men started finding the lanthanoids or rare earth elements in 1794 when yttrium was found by Johan Gadolin [1]. Later, the chemists analyzed the composition of yttrium and observed that it was contaminated with chemically similar elements [1]. As a result, two new elements terbium and erbium were isolated in 1843 [1].
Per Teodor Cleve started working on erbium in 1874 and was able to successfully isolate thulium in its oxide form in 1879 at the Uppsala University, Sweden [1, 5]. In 1911, the exact atomic weight of thulium was determined by Theodore William Richards, an American chemist who carried out 15,000 thulium bromate-recrystallizations to produce the pure form of thulium [1].
Thulium Identification |
|||
| Atomic number | 69 [1] | ||
| CAS number | 7440-30-4 [1] | ||
| Position in the periodic table | Group | Period | Block |
| Lanthanides [1] | 6 [1] | f [1] | |
Properties and Characteristics of Thulium
General Properties |
||||||||||||||||
| Relative atomic mass | 168.934 [1] | |||||||||||||||
Physical Properties |
||||||||||||||||
| Color | Bright silvery-gray [1, 5] | |||||||||||||||
| Melting point/freezing point | 1545 °C, 2813 °F [1] | |||||||||||||||
| Boiling point | 1950 °C, 3542 °F [1] | |||||||||||||||
| Density | 9.32 g cm-3 [1] | |||||||||||||||
| State of matter at room temperature (solid/liquid/gas) | Solid [1, 5] | |||||||||||||||
| Hardness | ||||||||||||||||
| – Brinell | 471 MPa [6] | |||||||||||||||
| – Mohs | 1.77 [6] | |||||||||||||||
| – Vickers | 520 MPa [6] | |||||||||||||||
| Electrical conductivity | 1.4X106 S/m [6] | |||||||||||||||
| Thermal (heat) conductivity | 17 W/(m K) [6] | |||||||||||||||
| Specific heat | 160 J kg-1 K-1 [1] | |||||||||||||||
| Bulk modulus | 44.5 GPa [1] | |||||||||||||||
| Shear modulus | 30.5 GPa [1] | |||||||||||||||
| Young’s modulus | 74 GPa [1] | |||||||||||||||
| Vapor pressure | ||||||||||||||||
| – Temperature (K) | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 | 2200 | 2400 | |||||
| – Pressure (Pa) | – | 6.03X 10-10 | 5.94X 10-5 | 5.61X 10-2 | 5.22 | 130 | – | – | – | – | – | |||||
Chemical Properties |
||||||||||||||||
| Oxidation state/Oxidation number | +2, +3, (+4) [1] | |||||||||||||||
| Isotopes | Isotope | Mass | Abundance | Half-life | Mode of decay | |||||||||||
| 169Tm | 168.934 | 100 | – | – | ||||||||||||
Atomic Data of Thulium (Element 69)
| Valence electrons | 3 [6] | |||||||
| Quantum numbers | 2F7/2 [6] | |||||||
| Electron configuration (noble gas configuration) | [Xe] 4f136s2 [1] | |||||||
| Atomic structure | ||||||||
| – Number of electrons | 69 [5] | |||||||
| – Number of neutrons | 100 [5] | |||||||
| – Number of protons | 69 [5] | |||||||
| Radius of Atom | ||||||||
| – Atomic radius | 2.27 Å [1] | |||||||
| – Covalent radius | 1.77 Å [1] | |||||||
| Electronegativity (Pauling-scale) | 1.25 [1] | |||||||
| Electron affinity | 99.283 kJ mol-1 [1] | |||||||
| Ionization energy (kJ mol-1) | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
| 596.695 | 1162.65 | 2284.77 | 4119.9 | – | – | – | – | |
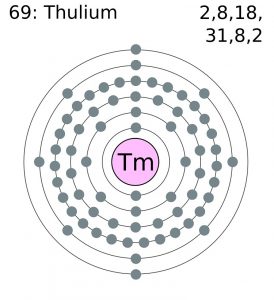
Thulium Electron Configuration (Bohr Model)
What is Thulium used for
- Thulium-170 (170Tm), a radioactive isotope formed by bombarding thulium in a nuclear reactor, can be used as a portable source of radiation in medical x-ray machines [1, 7].
- The element 69 is used as a dopant in fiber lasers for surgical applications [1, 3].
- Since it emits a bluish glow when exposed to ultra-violet light, thulium is used in Euro banknotes for detecting counterfeit currency [5].
- It can also be used with other lanthanides for producing alloys [5].
Possible Harmful Effects or Toxicity
Being a non-toxic element, it does not cause any health hazards [1].
Interesting Facts
- Because of its softness and malleability, thulium can be easily cut with a knife [3].
- The element is sometimes represented by an image of a snow-covered mountain to indicate the origin of its name, suggesting a region far off to the north [1].
Cost of Thulium
While one gram of thulium costs about $50 [8], a hundred gram of the element approximately costs $500-$700 [5, 8].
- References
- http://www.rsc.org/periodic-table/element/69/thulium
- https://education.jlab.org/itselemental/ele069.html
- https://www.livescience.com/38390-thulium.html
- https://www.webelements.com/thulium/isotopes.html
- https://www.chemicool.com/elements/thulium.html
- http://periodictable.com/Elements/069/data.html
- https://applets.kcvs.ca/IPTEI/pdf-elements/thulium.pdf
- https://www.radiochemistry.org/periodictable/elements/69.html