Yttrium
What is Yttrium
Denoted by the chemical symbol Y, yttrium (pronounced as IT-ree-em) belongs to the family of transition and lanthanide metals. It is considered to be one of the rare earth elements having 50 isotopes, with yttrium-89 being the only stable one [3, 9].
Where is Yttrium Found in Nature
It is found associated with other rare earth mineral ores like monazite and bastnaesite, in Canada, Brazil, Australia, Hungary, and Greece. The mineral ore of xenotime is another good source as it can constitute up to 50% of yttrium phosphate [1].
History
The element is named after a village in Ytterby, Sweden.
Who Discovered It
Finnish chemist and physicist Johan Gadolin discovered it in 1794.
When and Where was it Discovered
In 1787, the Swedish chemist, Karl Arrhenius sent an unusual black rock, found near a quarry at Ytterby in the Stockholm region, to Gadolin in Finland for detailed analysis. He assumed it to be some new tungsten mineral. In 1794, while testing the rock, Gadolin discovered the presence of a new oxide compound (later found to be yttrium oxide) that constituted 38% of its weight and named it ‘earth’ as it would not undergo further reduction after heating with charcoal [1].
In 1828, Friedrich Wohler isolated yttrium by reacting yttrium chloride with potassium. However, it contained other elements as well [1].
When Carl Mosander, in 1843, examined yttrium oxide thoroughly, he found two more oxides, namely of terbium and erbium, having a yellow and rose-red appearance respectively [1].
Classification and Position of the Element on the Periodic Table [1]
| Group | 3 |
| Period | 5 |
| Block | d |
Properties of Yttrium [1, 2, 3]
General Properties |
|
| Relative Atomic Mass | 88.906 |
Physical Properties |
|
| Color | Silver white |
| Luster | Metallic silver |
| Odor | Unknown |
| Malleability | Yes |
| Ductility | Yes |
| Melting Point/Freezing Point | 1522°C, 2772°F |
| Boiling Point | 3345°C, 6053°F |
| Density | 4.47 g/cm3 |
| State of Matter at Room Temperature (Normal Phase) | Solid |
| Work Function | 8.31 eV |
Chemical Properties |
|
| Flammability | Yes |
| Oxidation state/Oxidation number | (+1), (+2), +3 |
| Maximum Oxidation Number | 3 |
Atomic Data of Yttrium [1, 2, 3]
| Atomic Number | 39 | ||||||
| Valence Electrons | 4d 5s | ||||||
| Quantum Numbers | |||||||
| – n | 4 | ||||||
| – ℓ | 2 | ||||||
| – m ℓ | -2 | ||||||
| – m s | +1/2 | ||||||
| Ground State Electron Configuration | [Kr] 4d1 5s2 | ||||||
| Primary XPS Region | Y3d | ||||||
| Crystal Structure | hcp: hexagonal close-packed | ||||||
| Atomic Structure | |||||||
| – Number of Electrons | 39 | ||||||
| -Number of Neutrons | 50 | ||||||
| – Number of Protons | 39 | ||||||
| Radius of Atom | |||||||
| – Atomic Radius | 2.32 Å | ||||||
| – Covalent Radius | 1.76 Å | ||||||
| Electronegativity | 1.22 | ||||||
| Ionic Charge | +3 | ||||||
| Ionization Energy
(kJmol-1) |
1st | 2nd | 3rd | 4th | 5th | 6th | 7th |
| 599.878 | 1179.437 | 1979.88 | 5846.72 | 7429 | 8973 | 11192 | |
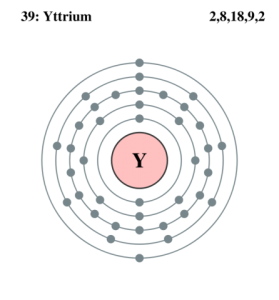
Yttrium Bohr Model
What is Yttrium Used for
- It is used to increase the strength of magnesium and aluminum alloys [3].
- Yttrium functions as an efficient catalyst in the polymerization of ethane [1].
- As a result of its high affinity for oxygen, the element is used as a deoxidizer in non-ferrous metals like vanadium [3].
- Yttrium is used in lithium iron phosphate batteries for increasing their capacity and durability [7].
- Yttrium-90, a radioisotope of Y, can be useful for radiation therapy for cancer treatment [1, 5], as well as in knee surgeries for treatment of osteoarthritis [8].
- Yttrium oxide is used in camera lenses, and various communication devices like radar and satellite [1, 3].
- Combining europium with yttrium oxide, yttrium orthovanadate, or yttrium oxysulfide produces red phosphorus, used in color television tubes [2, 3].
- Yttrium-aluminum-garnet (YAG) is used as a simulated diamond in jewelry-making [2]. Neodymium-doped YAG laser may also have several applications in the medical and scientific fields like in Lasik surgery and laser spectroscopy [6].

Yttrium Aluminum Garnet
Yttrium Toxicity and Health Hazards
While water-soluble compounds of yttrium exhibit mild toxicity, its insoluble compounds are believed to be harmless [3]. Exposure to yttrium compounds can have mild toxic effects and can cause symptoms like shortness of breath, chest pain, and coughing [10]. In case of extreme exposure, it might even lead to lung diseases. Animal studies show yttrium and its compounds to cause liver and lung damage [3].
Interesting Facts
- Analysis of rock and dust samples collected from the moon during the Apollo 11 mission has shown a high content of yttrium, ranging between 54 and 213 parts per million [3].
- The stability of Y is attributed to the formation of an oxide coating on its surface upon exposure to the oxygen in air [3].
Yttrium Cost
The price of pure yttrium ranges between $400 and $450 per 100 grams.
- References
- http://www.rsc.org/periodic-table/element/39/yttrium
- https://education.jlab.org/itselemental/ele039.html
- https://www.chemicool.com/elements/yttrium.html
- http://www.softschools.com/facts/periodic_table/yttrium_facts/215/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4640917/
- http://www.physics-and-radio-electronics.com/physics/laser/ndyaglaser.html
- https://medium.com/@balqon/lithium-yttrium-iron-phosphate-battery-abe28c943498
- https://www.ncbi.nlm.nih.gov/pubmed/19357547
- https://www.radiochemistry.org/periodictable/elements/39.html
- https://www.sciencedirect.com/science/article/pii/S2093791113410028