Reactivity Series
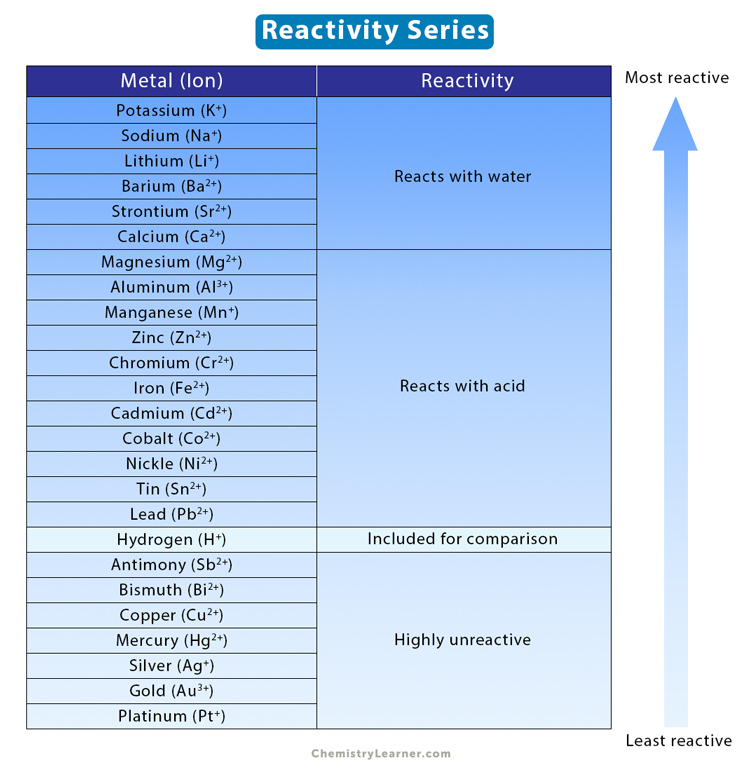
The reactivity series of metals is a list of metals arranged in their order of reactivity from highest to lowest. At the top of the list are the highly reactive metals that lose electrons during a chemical reaction to form ions. At the bottom are the least reactive metals. During a single-displacement reaction, a metal higher up the series will replace a lower metal in the series [1-4].
Origins
The reactivity series is due to the stability of the electron configuration of metals as ions. Metals donating only one electron to form stable ions are more reactive than similar metals donating more than one electron. It is for this reason that Group 1A metals or alkali metals are the most reactive [2].
Also, metals in higher periods tend to be more reactive as their outermost electrons (the ones that will be lost) exist far from the positively charged nucleus. Therefore, they are held less tightly.
Characteristics [4]
- Metals higher in the series displace lower metals from their salts during single-displacement reactions
- Metals at the top of the list, such as lithium, sodium, and potassium, are highly reactive and result in a vigorous reaction. They are also strong reducing agents and oxidize quickly. These metals tarnish and corrode easily.
- The reducing ability of metals decreases as we go down the series.
- Metals at the bottom of the chart, gold, silver, and platinum, usually are unreactive and do not react with acid and water.
- All metals above hydrogen liberate hydrogen gas (H2) upon reacting with dilute hydrochloric acid (HCl) or dilute sulfuric acid (H2SO4).
List
The list below represents the order of the reactivity series from most reactive to least reactive [4].
- Potassium
- Sodium
- Lithium
- Barium
- Radium
- Strontium
- Calcium
- Magnesium
- Beryllium
- Aluminum
- Titanium(IV)
- Manganese
- Zinc
- Chromium(III)
- Iron(II)
- Cadmium
- Cobalt(II)
- Nickel
- Tin
- Lead
- Antimony
- Bismuth(III)
- Copper(II)
- Tungsten
- Mercury
- Silver
- Gold
- Platinum
From this list, it is clear that potassium is the most reactive metal.
Significance
The reactivity series is essential for several reasons. It can predict the reactions between metals and water and metals and acids. Aside, it can predict the outcome of single-displacement reactions. It helps us understand why different metals are extracted from their ores differently. Based on the importance of the reactivity series, let us look at the various chemical reactions that metals undergo [1-4].
1. Reaction with Metallic Salts
Metals higher in the series tend to lose electrons and easily displace lower metals. This reaction is known as a single-displacement reaction. A characteristic of this reaction is that one cation trades places with another to form a new product.
Examples
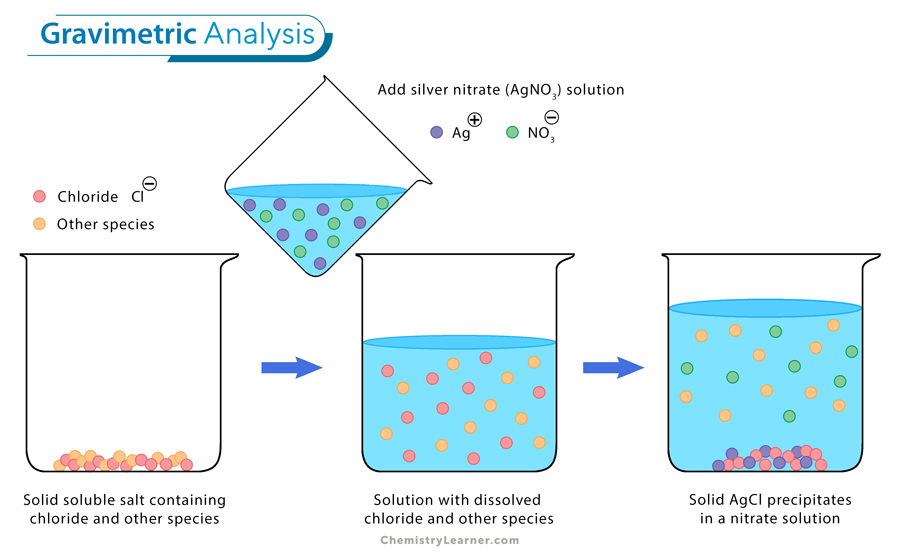
1. Copper (Cu) can displace silver (Ag) in an aqueous solution of silver nitrate (AgNO3).
Cu (s) + 2 AgNO3 (aq.) → Cu(NO3)2 (aq.) + 2 Ag (s/ppt.)
2. Magnesium (Mg) can displace copper (Cu) in an aqueous solution of copper nitrate (Cu(NO3)2).
Mg (s) + Cu(NO3)2 (aq.) → Mg(NO3)2 (aq.) + Cu (s/ppt.)
2. Reaction with Water
Highly reactive metals can react with cold water to form the corresponding hydroxide.
Examples
1. Sodium (Na) is highly reactive and displaces hydrogen (H2) from water (H2O).
2 Na (s) + 2 H2O (l) → 2 NaOH (aq.) + H2 (g)
2. Aluminum (Al) reacts vigorously with water to produce hydrogen gas.
2 Al (s) + 6 H2O (l) → 2 Al(OH)3 (aq.) + 3 H2 (g)
3. Reaction with Acid
Highly reactive metals produce hydrogen when reacting with acid. Each metal atom loses one or more electrons, and each acid molecule loses one or more protons. The electrons and protons combine to form a hydrogen atom. The amount of hydrogen produced depends on the starting mass of the metal and the acid’s strength. The reactivity of the metal determines the reaction rate. Metals that lose electrons easily produce a fast and vigorous reaction.
Examples
1. Zinc (Zn) reacts with sulfuric acid (H2SO4) to produce zinc sulfate (ZnSO4) while liberating hydrogen gas (H2).
Zn (s) + H2SO4 (aq.) → ZnSO4 (aq.) + H2 (g)
2. Iron will react with sulfuric acid to give hydrogen and iron (II) sulfate.
Fe (s) + H2SO4 (l) → FeSO4 (aq.) + H2 (g)
The following chart summarizes the reactions discussed above [2].
Thus, the alkali metals are the most reactive, followed by the alkaline earth and transition metals. Noble metals like gold, silver, and platinum are not very reactive. Metals from potassium to calcium are highly reactive and react with cold water. On the other hand, metals from magnesium to lead react with acid. Metals from antimony to platinum are highly unreactive.
FAQs
Ans. Although it is not a metal, hydrogen has some characteristics that make it behave like alkali metals in some chemical reactions. It has only one electron. So, its electron configuration resembles that of other alkali metals.
Ans. Aluminum is a reactive metal and reacts with acid. On exposure to air, aluminum forms a hard protective layer of aluminum oxide (Al2O3), which prevents further oxidation.